QUESTION IMAGE
Question
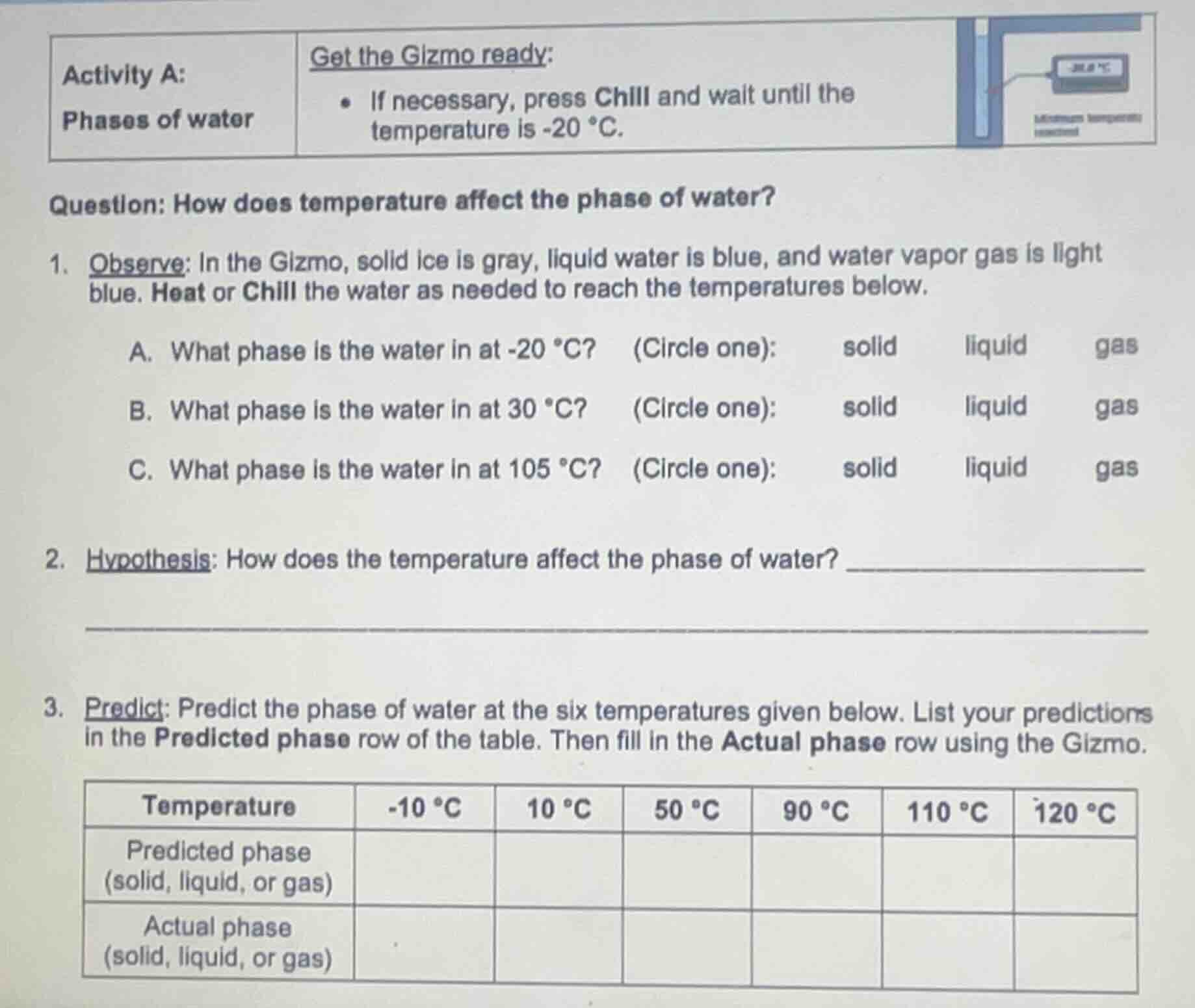
activity a:
phases of water
get the gizmo ready:
- if necessary, press chill and wait until the temperature is -20 °c.
question: how does temperature affect the phase of water?
- observe: in the gizmo, solid ice is gray, liquid water is blue, and water vapor gas is light blue. heat or chill the water as needed to reach the temperatures below.
a. what phase is the water in at -20 °c? (circle one): solid liquid gas
b. what phase is the water in at 30 °c? (circle one): solid liquid gas
c. what phase is the water in at 105 °c? (circle one): solid liquid gas
- hypothesis: how does the temperature affect the phase of water?
- predict: predict the phase of water at the six temperatures given below. list your predictions in the predicted phase row of the table. then fill in the actual phase row using the gizmo.
| temperature | -10 °c | 10 °c | 50 °c | 90 °c | 110 °c | 120 °c |
|---|---|---|---|---|---|---|
| actual phase (solid, liquid, or gas) |
Brief Explanations
- For the observation questions: Water freezes at 0 °C, so below this temperature it is solid; between 0 °C and 100 °C it is liquid; above 100 °C (at standard pressure) it is gas.
- The hypothesis is based on standard phase change behavior of water with temperature changes.
- Predictions follow the same phase change rules, and actual phases match these predictions under standard atmospheric pressure.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.
A. solid
B. liquid
C. gas
- As temperature increases, water changes from solid (ice) to liquid (water) to gas (water vapor); as temperature decreases, it changes from gas to liquid to solid, at standard atmospheric pressure.
3.
| Temperature | -10 °C | 10 °C | 50 °C | 90 °C | 110 °C | 120 °C |
|---|---|---|---|---|---|---|
| Actual phase (solid, liquid, or gas) | solid | liquid | liquid | liquid | gas | gas |