QUESTION IMAGE
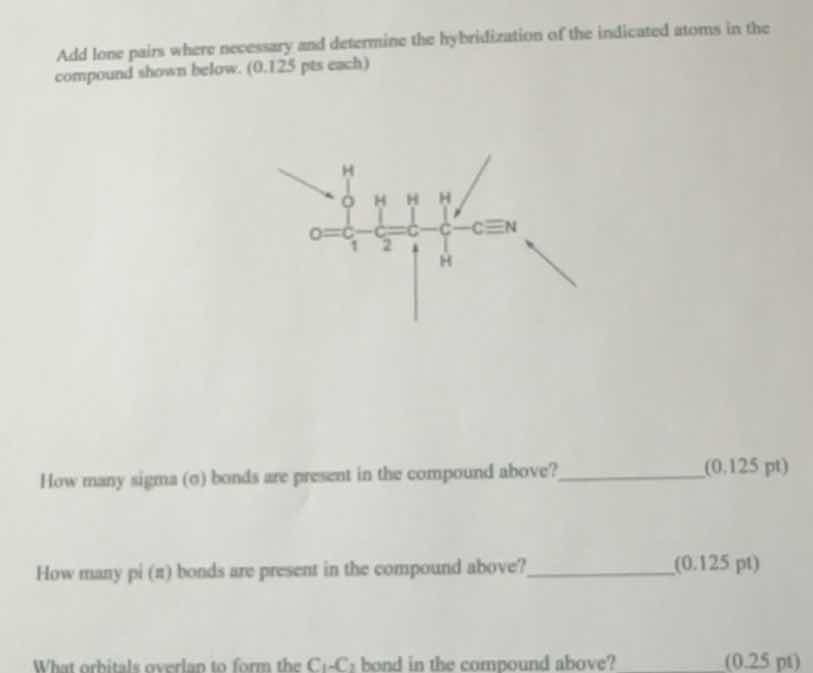
Question
add lone pairs where necessary and determine the hybridization of the indicated atoms in the compound shown below. (0.125 pts each)
how many sigma (σ) bonds are present in the compound above? ____________ (0.125 pt)
how many pi (π) bonds are present in the compound above? ____________ (0.125 pt)
what orbitals overlap to form the c₁ - c₂ bond in the compound above? ________ (0.25 pt)
Step1: Add lone pairs to atoms
Oxygen has 2 lone pairs: $\overset{\Large{..}}{O}=C_1$; Nitrogen has 1 lone pair: $C\equiv\overset{\Large{..}}{N}$
Step2: Calculate hybridization for C₁
C₁ has 3 electron domains (double bond to O, single bond to H, single bond to C₂). Hybridization: $sp^2$
Step3: Calculate hybridization for C₂
C₂ has 4 electron domains (two single bonds to H, single bonds to C₁ and C₃). Hybridization: $sp^3$
Step4: Calculate hybridization for C₃
C₃ has 3 electron domains (two single bonds to H, single bond to C₂, single bond to C₄). Hybridization: $sp^3$
Step5: Calculate hybridization for C₄
C₄ has 2 electron domains (single bond to H, single bond to C₃, triple bond to N). Hybridization: $sp$
Step6: Count sigma (σ) bonds
Each single bond = 1 σ; double bond = 1 σ; triple bond = 1 σ. Total: $1(\text{O-H}) + 1(\text{O-C}_1) + 1(\text{C}_1\text{-C}_2) + 2(\text{C}_2\text{-H}) + 1(\text{C}_2\text{-C}_3) + 2(\text{C}_3\text{-H}) + 1(\text{C}_3\text{-C}_4) + 1(\text{C}_4\text{-H}) + 1(\text{C}_4\text{-N}) = 11$
Step7: Count pi (π) bonds
Double bond = 1 π; triple bond = 2 π. Total: $1(\text{O=C}_1) + 2(\text{C}_4\equiv\text{N}) = 3$
Step8: Identify C₁-C₂ overlapping orbitals
C₁ is $sp^2$ hybridized, C₂ is $sp^3$ hybridized. Bond forms from $\boldsymbol{sp^2-sp^3}$ overlap.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Lone pairs added: Oxygen (2 lone pairs), Nitrogen (1 lone pair)
- Hybridizations:
- C₁: $sp^2$
- C₂: $sp^3$
- C₃: $sp^3$
- C₄: $sp$
- Number of sigma (σ) bonds: 11
- Number of pi (π) bonds: 3
- Orbitals for C₁-C₂ bond: $sp^2$ orbital of C₁ and $sp^3$ orbital of C₂