QUESTION IMAGE
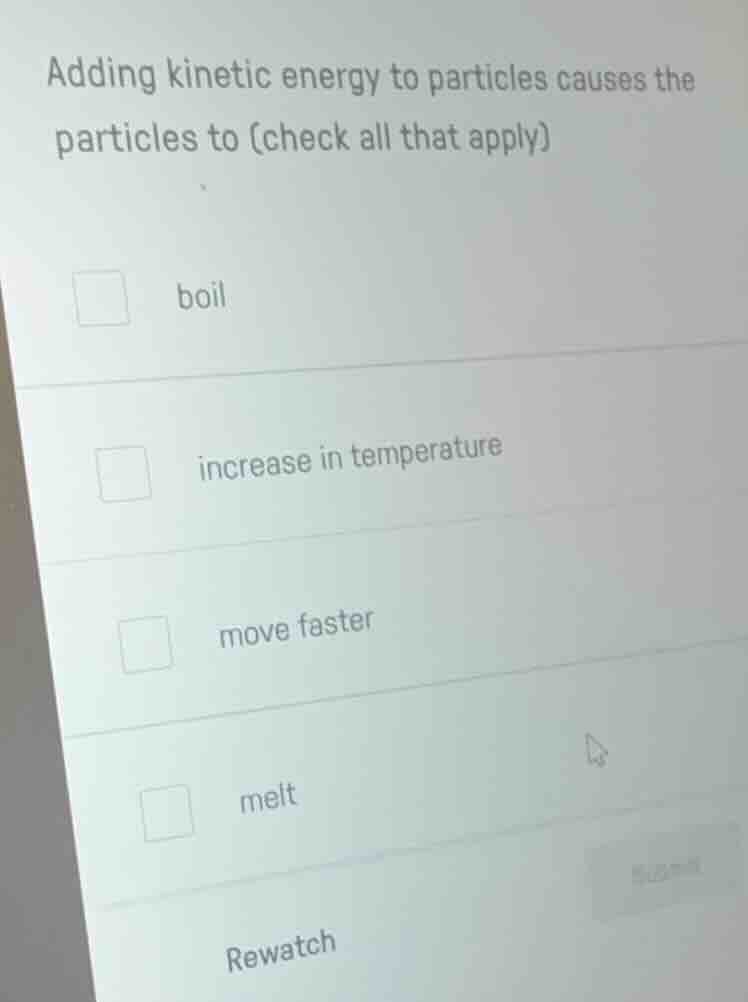
Question
adding kinetic energy to particles causes the particles to (check all that apply)
boil
increase in temperature
move faster
melt
rewatch
Brief Explanations
Kinetic energy relates to particle motion; adding it directly makes particles move faster. For most substances, increased particle motion leads to a temperature rise. Boiling and melting are phase changes that require adding potential energy (not just kinetic) at a constant temperature, so they are not direct results of adding kinetic energy alone.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- increase in temperature
- move faster