QUESTION IMAGE
Question
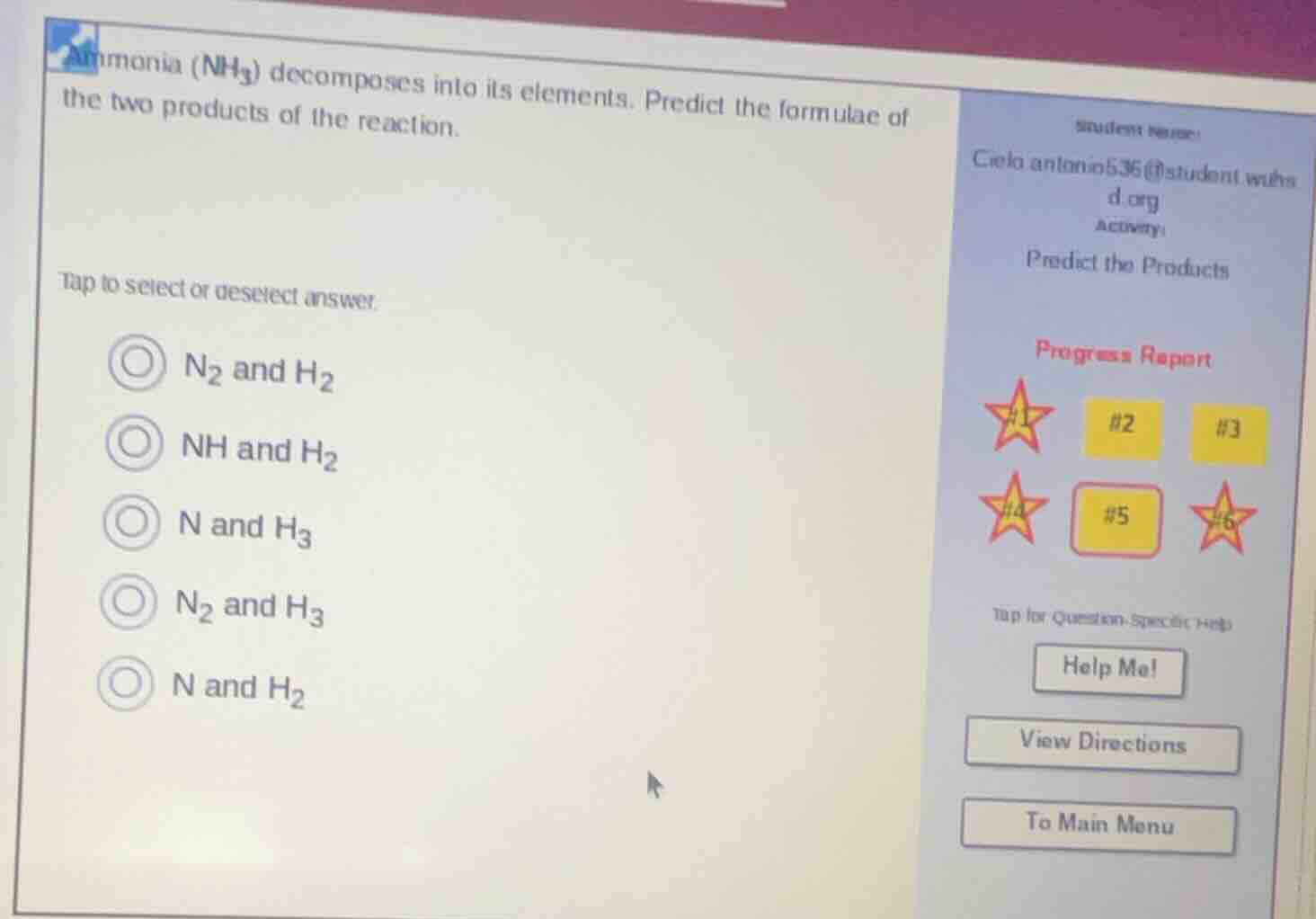
ammonia ($nh_3$) decomposes into its elements. predict the formulae of the two products of the reaction. tap to select or deselect answer. $n_2$ and $h_2$; $nh$ and $h_2$; $n$ and $h_3$; $n_2$ and $h_3$; $n$ and $h_2$
Ammonia ($\ce{NH_3}$) decomposes into its constituent elements. The elements in ammonia are nitrogen (N) and hydrogen (H). Nitrogen is a diatomic gas in its elemental form, so it exists as $\ce{N_2}$, and hydrogen is also diatomic, existing as $\ce{H_2}$. So the products of the decomposition of $\ce{NH_3}$ into its elements should be $\ce{N_2}$ and $\ce{H_2}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\ce{N_2}$ and $\ce{H_2}$