QUESTION IMAGE
Question
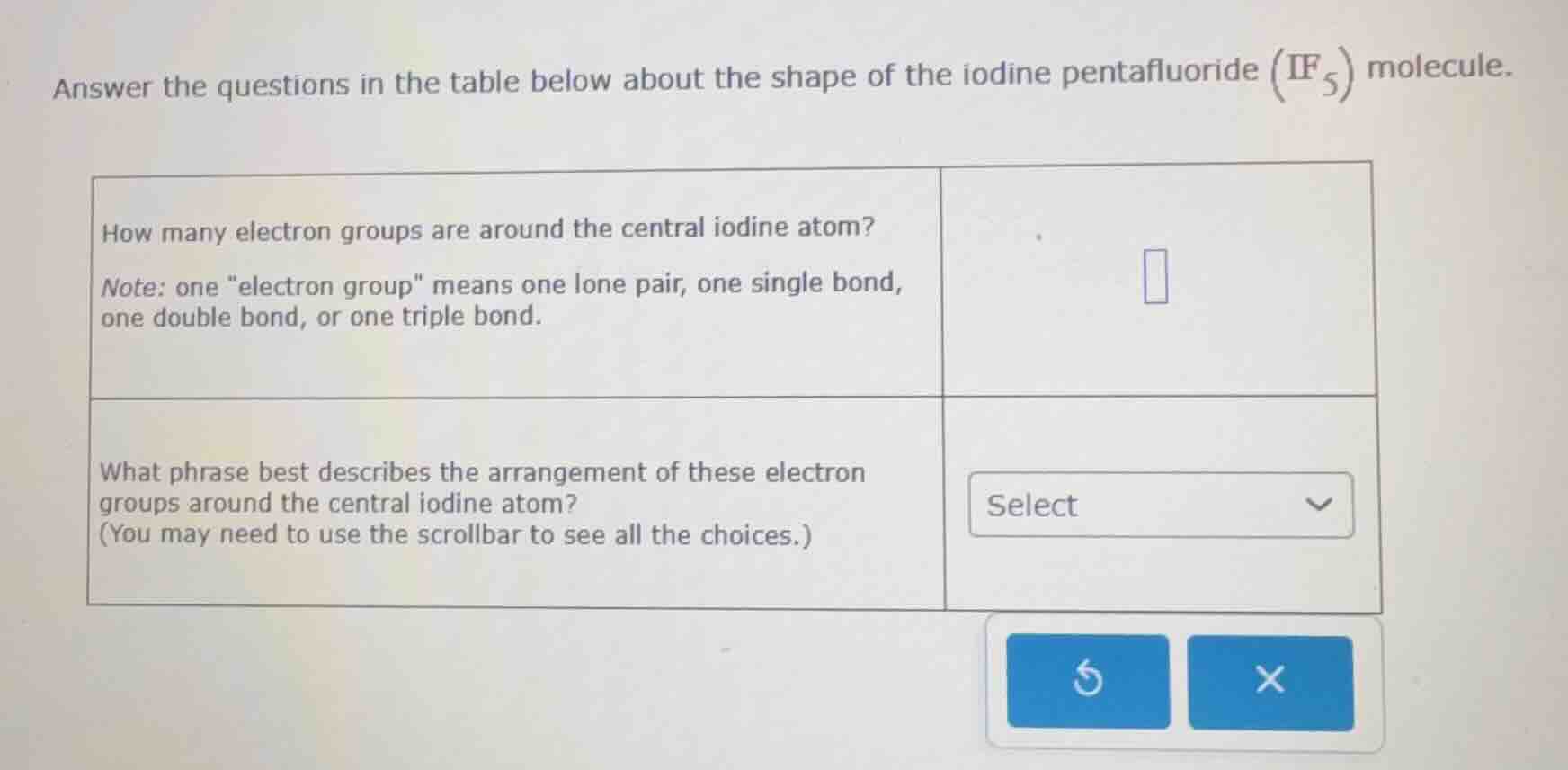
answer the questions in the table below about the shape of the iodine pentafluoride \\(\ce{if5}\\) molecule.
| how many electron groups are around the central iodine atom?
note: one \electron group\ means one lone pair, one single bond,
one double bond, or one triple bond. | \\(\square\\) |
| what phrase best describes the arrangement of these electron
groups around the central iodine atom?
(you may need to use the scrollbar to see all the choices.) | select |
First Question (Number of Electron Groups)
Step1: Determine valence electrons of I
Iodine (I) is in group 17, so it has 7 valence electrons. Fluorine (F) is also in group 17, each has 7 valence electrons. In $\ce{IF5}$, there are 5 F atoms bonded to I.
Step2: Calculate bonding and lone pairs
The number of bonding electrons: Each I - F bond is a single bond, so 5 bonds mean 5 electron groups from bonding. Now, calculate the lone pair on I: Total valence electrons for $\ce{IF5}$: $7 + 5\times7 = 7 + 35 = 42$. Electrons used in bonding: $5\times2 = 10$ (each single bond has 2 electrons). Remaining electrons: $42 - 10 = 32$. But these are around I? Wait, no, the formula for electron groups: For the central atom (I), the number of electron groups is the number of bonding groups (single/double/triple bonds) plus the number of lone pairs. The steric number (electron groups) is calculated as: number of bonded atoms + number of lone pairs. To find lone pairs on I: Valence electrons of I: 7. Electrons used in bonding: 5 (each bond uses 1 electron from I? Wait, no, in a single bond, each atom contributes 1 electron. So I contributes 5 electrons for 5 bonds, so remaining electrons on I: $7 - 5 = 2$? Wait, no, the correct way is: The formula for formal charge is not needed here. The steric number (electron groups) for I in $\ce{IF5}$: The central I has 5 bonding groups (5 I - F single bonds) and 1 lone pair (because $7 - 5 = 2$? Wait, no, let's use the VSEPR formula. The number of electron groups (steric number) is calculated as: number of atoms bonded to central atom + number of lone pairs on central atom. To find lone pairs on central I: The total number of valence electrons is $7 (I) + 5\times7 (F) = 42$. The number of electrons used in bonding: $5\times2 = 10$ (each single bond has 2 electrons). The remaining electrons: $42 - 10 = 32$, but these are distributed as lone pairs on F? No, F is a halogen, it has 7 valence electrons, so in a single bond, F has 6 lone pair electrons (since it shares 1 with I). So the lone pair on I: Let's use the formula for lone pairs on central atom: $LP = \frac{V - N - B}{2}$, where $V$ is valence electrons of central atom, $N$ is number of atoms bonded, $B$ is number of bonds (each bond is 1 for single, 2 for double, etc.). So $V = 7$, $N = 5$, $B = 5$ (single bonds). So $LP = \frac{7 - 5 - 5}{2}$? Wait, that can't be. Wait, no, the correct formula is $LP = \frac{V - (N\times1) - (B\times1)}{2}$? No, I think I messed up. Let's look up $\ce{IF5}$ structure. $\ce{IF5}$ has a square pyramidal shape. The central I has 5 bonding groups (5 F atoms) and 1 lone pair. So the number of electron groups is $5 + 1 = 6$. Ah, right! So the steric number (electron groups) is 6. So the first answer is 6.
Step2: Confirm
So the central I has 5 single bonds (5 electron groups from bonding) and 1 lone pair (1 electron group from lone pair), so total electron groups: $5 + 1 = 6$.
The electron groups around the central I atom in $\ce{IF5}$ are 6 (5 bonding, 1 lone pair). The VSEPR theory states that for a steric number of 6, the electron group arrangement (electron - pair geometry) is octahedral. Because octahedral geometry is the arrangement when there are 6 electron groups (lone pairs or bonding groups) around a central atom, with bond angles of approximately 90° between adjacent groups.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(First Question): 6