QUESTION IMAGE
Question
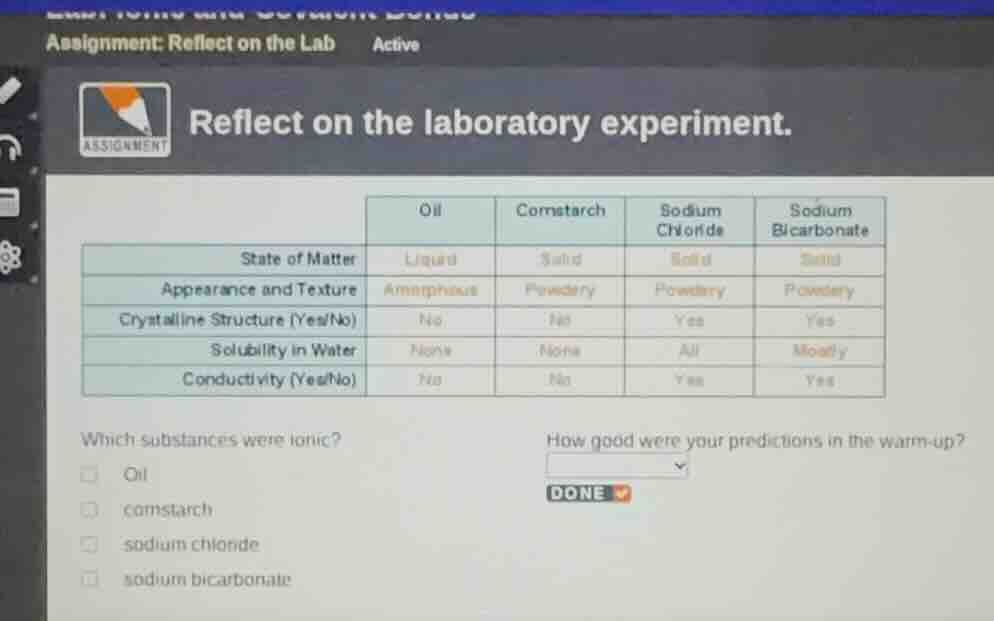
assignment: reflect on the lab active reflect on the laboratory experiment. oil cornstarch sodium chloride sodium bicarbonate state of matter liquid solid solid solid appearance and texture amorphous powdery powdery powdery crystalline structure (yes/no) no no yes yes solubility in water none none all mostly conductivity (yes/no) no no yes yes which substances were ionic? oil cornstarch sodium chloride sodium bicarbonate how good were your predictions in the warm - up? done
Ionic substances typically form crystalline structures, are soluble in water, and conduct electricity when dissolved (shown by "Yes" for conductivity here). From the table, sodium chloride and sodium bicarbonate meet these criteria: they have a crystalline structure, are soluble in water, and conduct electricity. Oil and cornstarch lack these ionic properties.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- sodium chloride
- sodium bicarbonate