QUESTION IMAGE
Question
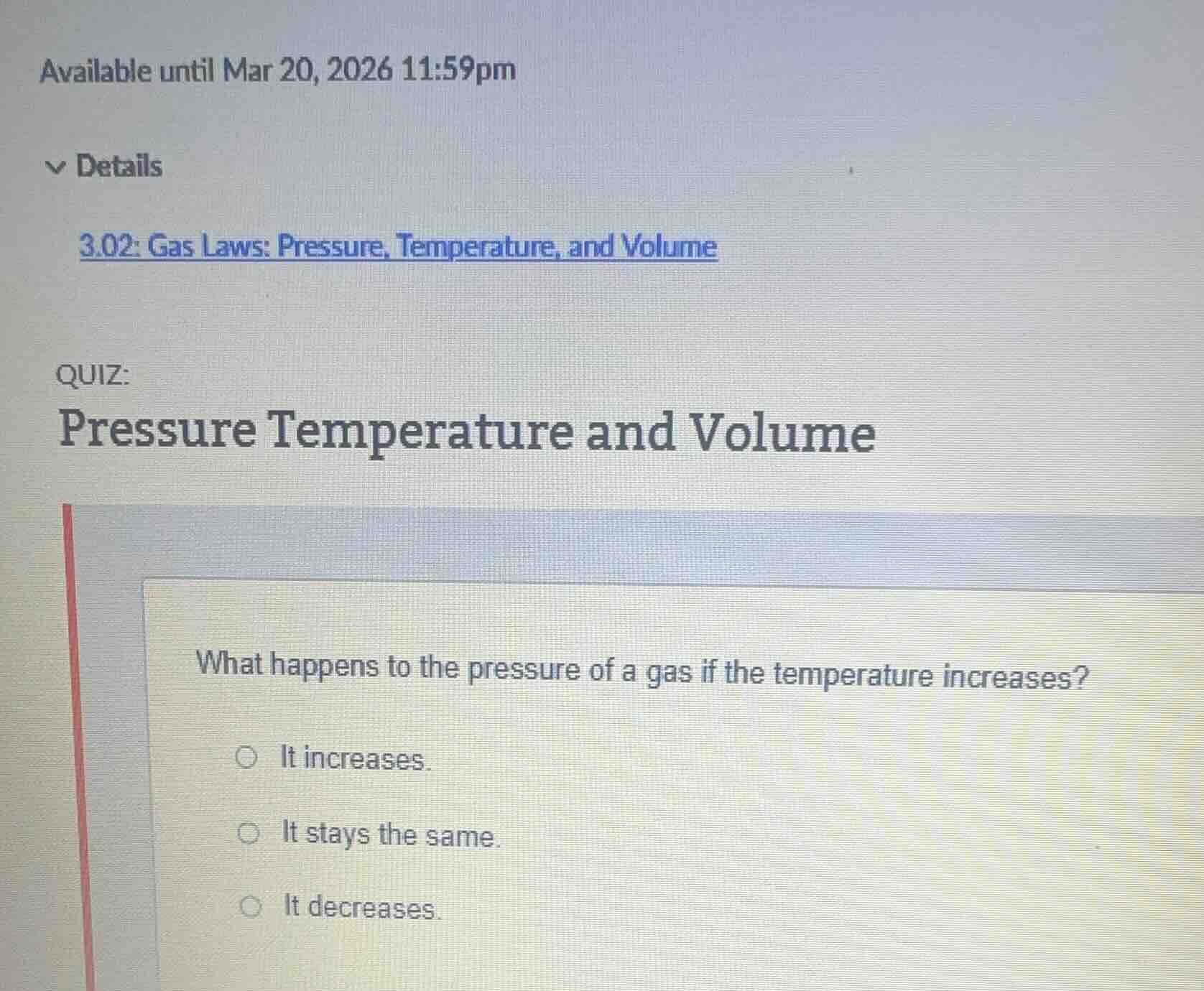
available until mar 20, 2026 11:59pm
details
3.02: gas laws: pressure, temperature, and volume
quiz:
pressure temperature and volume
what happens to the pressure of a gas if the temperature increases?
○ it increases.
○ it stays the same.
○ it decreases.
This is based on Gay-Lussac's Law (or the pressure-temperature law for gases), which states that for a fixed amount of gas at constant volume, pressure and absolute temperature are directly proportional. As temperature increases, gas molecules move faster, colliding with the container walls more forcefully and frequently, increasing pressure.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. It increases.