QUESTION IMAGE
Question
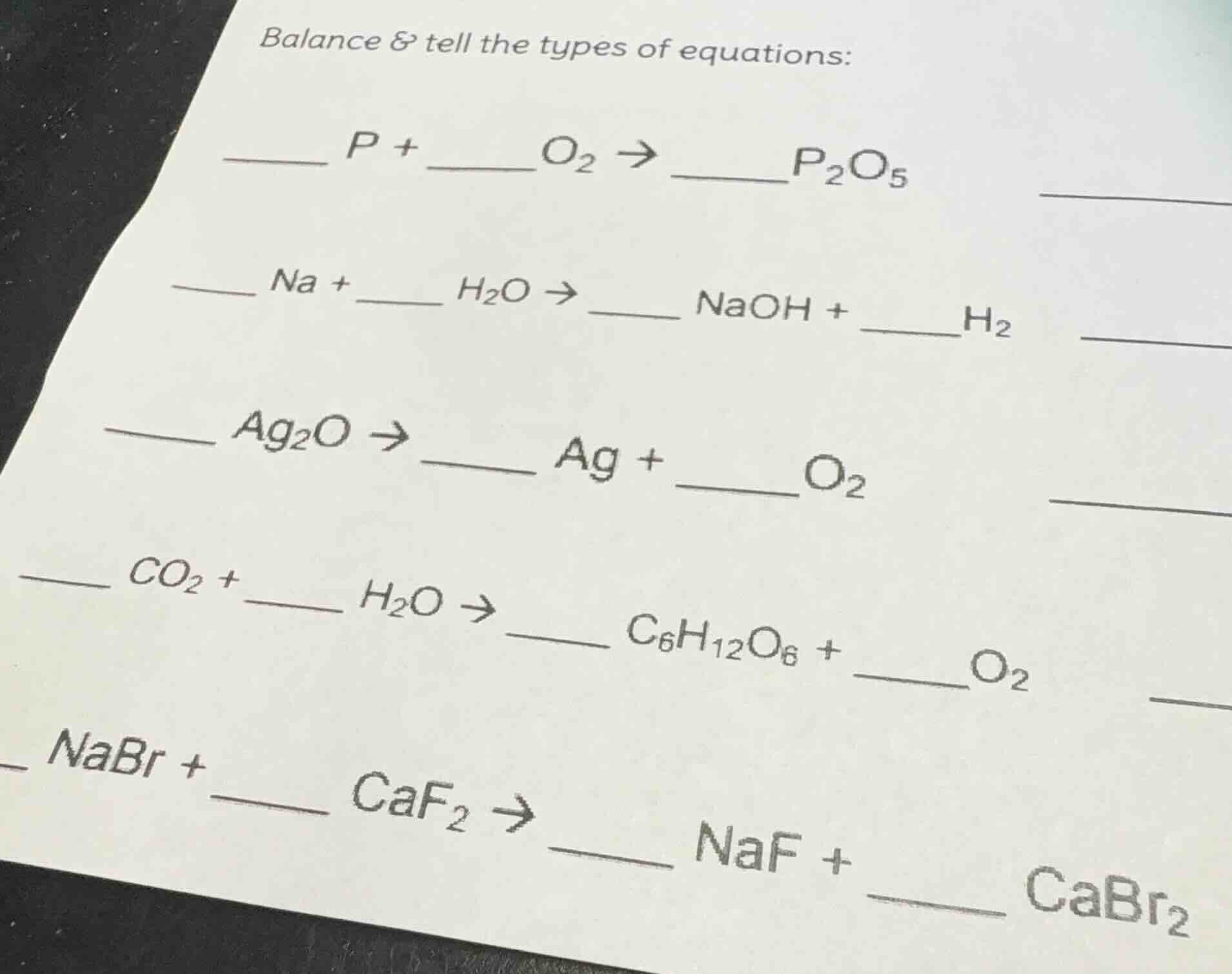
balance & tell the types of equations:
___ p + _o₂ → ___p₂o₅
___ na + _ h₂o → _ naoh + ___h₂
___ ag₂o → _ ag + ___o₂
___ co₂ + _ h₂o → _ c₆h₁₂o₆ + ___o₂
___ nabr + _ caf₂ → _ naf + ___ cabr₂
First Equation: $\boldsymbol{P + O_2
ightarrow P_2O_5}$
Step1: Balance O atoms
On right, $P_2O_5$ has 5 O. Left has $O_2$. Find LCM of 2 and 5, which is 10. So put 5 in front of $O_2$ (5×2=10 O) and 2 in front of $P_2O_5$ (2×5=10 O).
Now equation: $P + 5O_2
ightarrow 2P_2O_5$
Step2: Balance P atoms
Right has 2×2 = 4 P. So put 4 in front of P.
Balanced: $4P + 5O_2
ightarrow 2P_2O_5$
Step3: Identify reaction type
Two substances (P, $O_2$) combine to form one ($P_2O_5$) → Synthesis Reaction
Second Equation: $\boldsymbol{Na + H_2O
ightarrow NaOH + H_2}$
Step1: Balance H atoms
Right: $NaOH$ has 1 H, $H_2$ has 2 → total 3? Wait, better: Left $H_2O$ has 2 H per molecule. Let's balance O first (only in $H_2O$ and $NaOH$). $NaOH$ has 1 O, $H_2O$ has 1 O. So balance Na and O together. Let's try putting 2 in front of $Na$, $H_2O$, $NaOH$.
Now: $2Na + 2H_2O
ightarrow 2NaOH + H_2$
Step2: Check H
Left: 2×2 = 4 H. Right: 2×1 (from NaOH) + 2 (from $H_2$) = 4 H. Balanced.
Step3: Reaction type
$Na$ displaces H from $H_2O$ → Single Displacement (Metal + Water → Base + $H_2$)
Third Equation: $\boldsymbol{Ag_2O
ightarrow Ag + O_2}$
Step1: Balance O atoms
Left: $Ag_2O$ has 1 O. Right: $O_2$ has 2. Put 2 in front of $Ag_2O$ (2 O) and 1 in front of $O_2$ (2 O? Wait, 2×1=2 O, 1×2=2 O. Now: $2Ag_2O
ightarrow Ag + O_2$
Step2: Balance Ag atoms
Left: 2×2 = 4 Ag. So put 4 in front of Ag.
Balanced: $2Ag_2O
ightarrow 4Ag + O_2$
Step3: Reaction type
One compound breaks into two (Ag and $O_2$) → Decomposition Reaction
Fourth Equation: $\boldsymbol{CO_2 + H_2O
ightarrow C_6H_{12}O_6 + O_2}$ (Photosynthesis)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $4P + 5O_2
ightarrow 2P_2O_5$ (Synthesis)
- $2Na + 2H_2O
ightarrow 2NaOH + H_2$ (Single Displacement)
- $2Ag_2O
ightarrow 4Ag + O_2$ (Decomposition)
- $6CO_2 + 6H_2O
ightarrow C_6H_{12}O_6 + 6O_2$ (Synthesis/Photosynthesis)
- $2NaBr + CaF_2
ightarrow 2NaF + CaBr_2$ (Double Displacement)