QUESTION IMAGE
Question
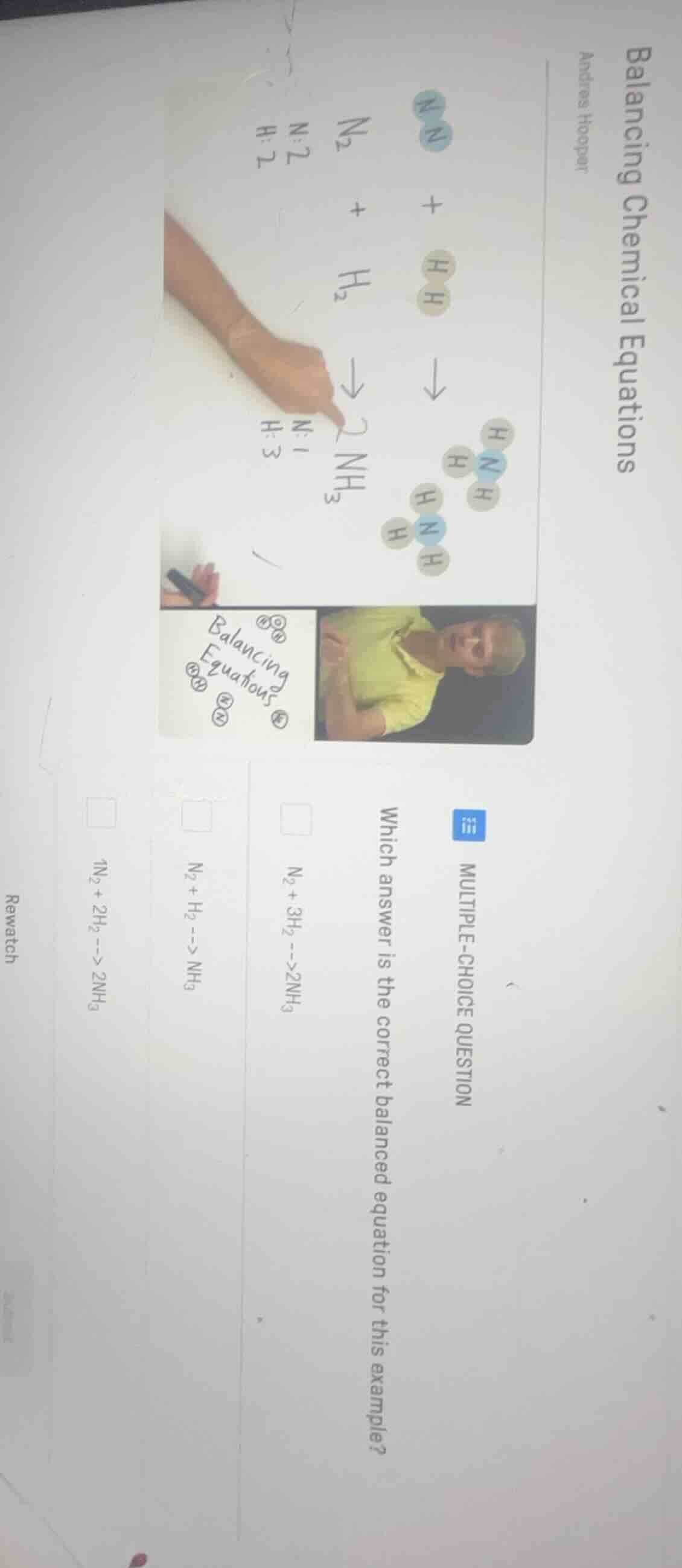
balancing chemical equations andres hooper multiple-choice question which answer is the correct balanced equation for this example? $\ce{n_{2} + 3h_{2} -> 2nh_{3}}$ $\ce{n_{2} + h_{2} -> nh_{3}}$ $\ce{n_{2} + 2h_{2} -> 2nh_{3}}$ rewatch
Step1: Count N atoms on each side
Reactants: $\text{N}_2$ has 2 N atoms. Products: $\text{NH}_3$ has 1 N atom each, so use coefficient 2 for $\text{NH}_3$ to get 2 N atoms: $\text{N}_2 + \text{H}_2
ightarrow 2\text{NH}_3$
Step2: Count H atoms on each side
Products: $2\text{NH}_3$ has $2\times3=6$ H atoms. Reactants: $\text{H}_2$ has 2 H atoms, so use coefficient 3 for $\text{H}_2$ to get $3\times2=6$ H atoms: $\text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3$
Step3: Verify atom balance
N: 2 on left, $2\times1=2$ on right. H: $3\times2=6$ on left, $2\times3=6$ on right. Equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3$ (the first multiple-choice option)