QUESTION IMAGE
Question
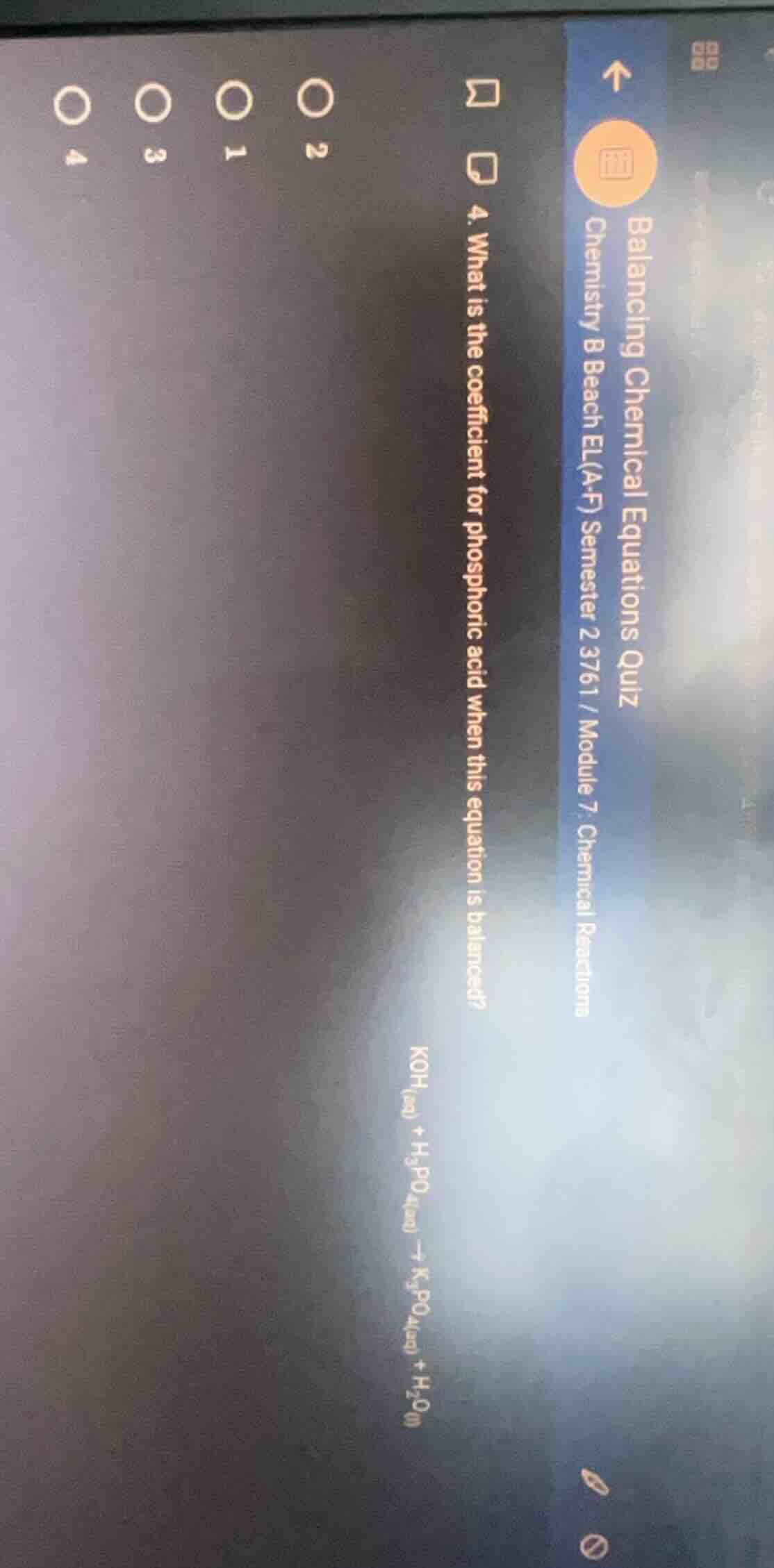
balancing chemical equations quiz chemistry b beach el(a-f) semester 2 3761 / module 7: chemical reactions 4. what is the coefficient for phosphoric acid when this equation is balanced? koh(aq) + h₃po₄(aq) → k₃po₄(aq) + h₂o(l)
Step1: List the atoms on each side
Reactants: K, O, H, P. Products: K, O, H, P.
- K: 1 (left), 3 (right)
- P: 1 (left), 1 (right)
- O: 1 (KOH) + 4 (H₃PO₄) = 5 (left), 4 (K₃PO₄) + 1 (H₂O) = 5 (but need to balance K first, then H and O)
- H: 1 (KOH) + 3 (H₃PO₄) = 4 (left), 2 (H₂O) (but will change with K)
Step2: Balance K
To balance K, put coefficient 3 in front of KOH: \( 3KOH_{(aq)} + H_3PO_{4(aq)}
ightarrow K_3PO_{4(aq)} + H_2O_{(l)} \)
Now K: 3 (left), 3 (right)
Step3: Balance H
Left H: 3 (from 3KOH) + 3 (from H₃PO₄) = 6. Right H: 2 (from H₂O). So put coefficient 3 in front of H₂O: \( 3KOH_{(aq)} + H_3PO_{4(aq)}
ightarrow K_3PO_{4(aq)} + 3H_2O_{(l)} \)
Now H: 6 (left), 6 (right)
Step4: Check O
Left O: 3 (from 3KOH) + 4 (from H₃PO₄) = 7. Right O: 4 (from K₃PO₄) + 3 (from 3H₂O) = 7. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficient for phosphoric acid (\(H_3PO_4\)) is 1. Wait, no, wait. Wait, in the balanced equation: \(3KOH + H_3PO_4
ightarrow K_3PO_4 + 3H_2O\). Wait, no, wait, let's recheck. Wait, KOH is 3, H₃PO₄ is 1, K₃PO₄ is 1, H₂O is 3. So the coefficient for \(H_3PO_4\) is 1? Wait, no, maybe I made a mistake. Wait, original equation: \(KOH + H_3PO_4
ightarrow K_3PO_4 + H_2O\). Balancing K: 3 KOH. Then H: 3 KOH has 3 H, H₃PO₄ has 3 H, total 6 H. So H₂O needs 3 (3*2=6). Then O: 3 KOH has 3 O, H₃PO₄ has 4 O, total 7 O. K₃PO₄ has 4 O, 3 H₂O has 3 O, total 7 O. So yes, the coefficient for \(H_3PO_4\) is 1? Wait, but the options are 4,3,1,2. Wait, maybe the equation was \(KOH + H_3PO_4
ightarrow K_3PO_4 + H_2O\). So when balanced, it's \(3KOH + H_3PO_4 = K_3PO_4 + 3H_2O\). So the coefficient for \(H_3PO_4\) is 1. But wait, maybe the original equation was different? Wait, the user's equation: \(KOH_{(aq)} + H_3PO_{4(aq)}
ightarrow K_3PO_{4(aq)} + H_2O_{(l)}\). So balancing:
- K: 1 vs 3 → put 3 in KOH: \(3KOH + H_3PO_4
ightarrow K_3PO_4 + H_2O\)
- P: 1 vs 1 → ok.
- H: 3 (from 3KOH) + 3 (from H₃PO₄) = 6 → H₂O needs 3 (3*2=6): \(3KOH + H_3PO_4
ightarrow K_3PO_4 + 3H_2O\)
- O: 3 (3KOH) + 4 (H₃PO₄) = 7; 4 (K_3PO4) + 3 (3H2O) = 7 → balanced.
So the coefficient for \(H_3PO_4\) is 1. But the options are 4,3,1,2. So the answer is 1? Wait, but maybe I misread the equation. Wait, maybe the phosphoric acid is \(H_3PO_4\) (correct formula) and the product is \(K_3PO_4\). So yes, the coefficient is 1. Wait, but let me check again.
Wait, another way: count each atom:
After putting 3 in KOH and 1 in H₃PO₄, 1 in K₃PO₄, 3 in H₂O:
- K: 3 (31) = 3 (13) ✔️
- P: 1 (11) = 1 (11) ✔️
- O: 3 (31) + 4 (14) = 7; 4 (14) + 3 (31) = 7 ✔️
- H: 3 (31) + 3 (13) = 6; 6 (3*2) = 6 ✔️
Yes, so the coefficient for \(H_3PO_4\) is 1. Wait, but the options are 4,3,1,2. So the answer is 1. Wait, but maybe the equation was written as \(KOH + H_3PO_4
ightarrow K_3PO_4 + H_2O\) and the coefficient for \(H_3PO_4\) is 1. So the answer is 1.