QUESTION IMAGE
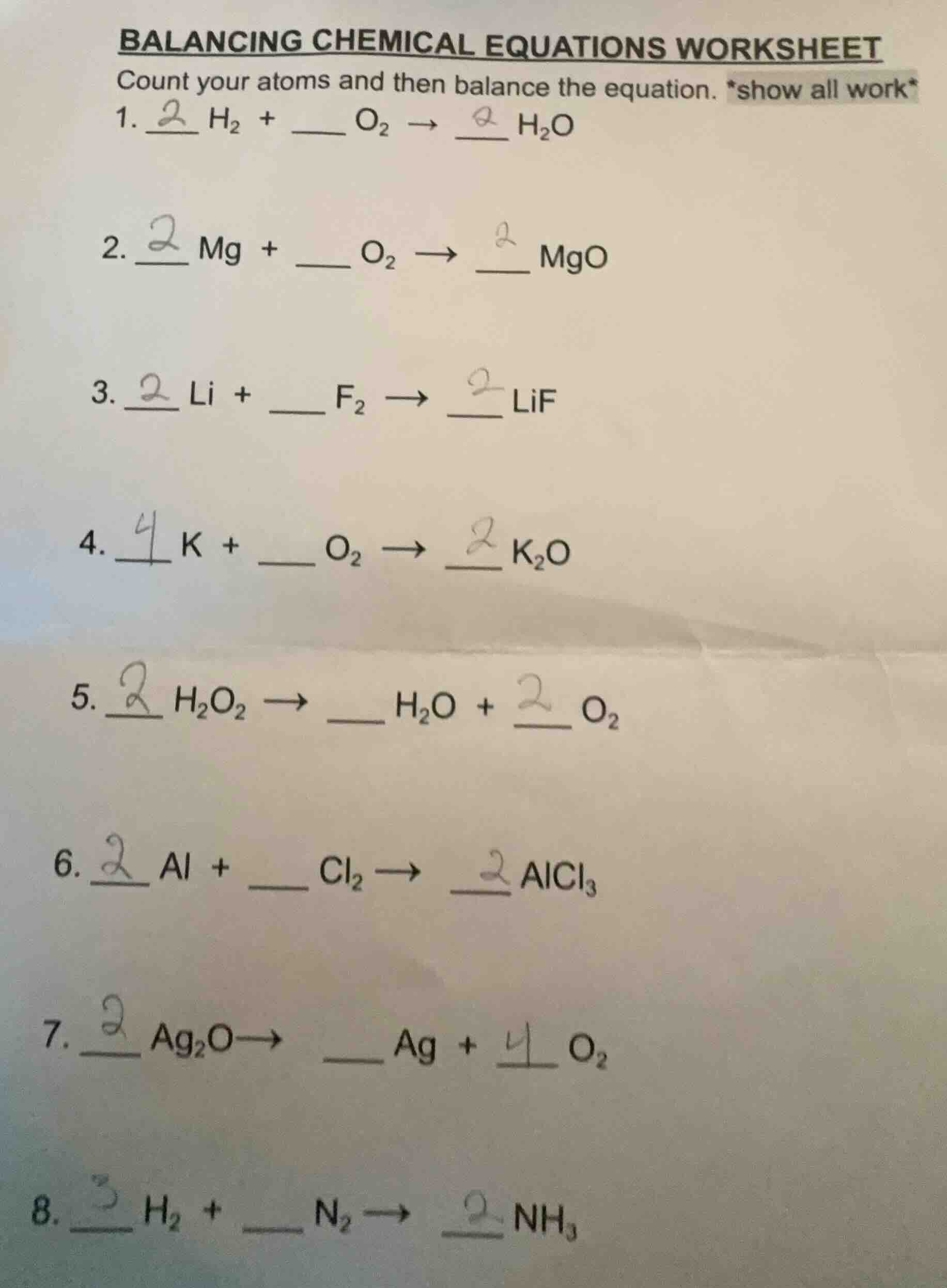
Question
balancing chemical equations worksheet
count your atoms and then balance the equation. show all work
- $\boldsymbol{2}$ $ce{h_{2}}$ + ____ $ce{o_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{h_{2}o}$
- $\boldsymbol{2}$ $ce{mg}$ + ____ $ce{o_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{mgo}$
- $\boldsymbol{2}$ $ce{li}$ + ____ $ce{f_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{lif}$
- $\boldsymbol{4}$ $ce{k}$ + ____ $ce{o_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{k_{2}o}$
- $\boldsymbol{2}$ $ce{h_{2}o_{2}}$ $
ightarrow$ ____ $ce{h_{2}o}$ + $\boldsymbol{2}$ $ce{o_{2}}$
- $\boldsymbol{2}$ $ce{al}$ + ____ $ce{cl_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{alcl_{3}}$
- $\boldsymbol{2}$ $ce{ag_{2}o}$$
ightarrow$ ____ $ce{ag}$ + $\boldsymbol{4}$ $ce{o_{2}}$
- $\boldsymbol{3}$ $ce{h_{2}}$ + ____ $ce{n_{2}}$ $
ightarrow$ $\boldsymbol{2}$ $ce{nh_{3}}$
Step1: Balance O atoms (Eq1)
Initial: $\ce{2H2 + _O2 -> 2H2O}$; O: 2 on left, 2 on right → coefficient 1 for $\ce{O2}$
Step2: Balance O atoms (Eq2)
Initial: $\ce{2Mg + _O2 -> 2MgO}$; O: 2 on left, 2 on right → coefficient 1 for $\ce{O2}$
Step3: Balance F atoms (Eq3)
Initial: $\ce{2Li + _F2 -> 2LiF}$; F: 2 on left, 2 on right → coefficient 1 for $\ce{F2}$
Step4: Balance O atoms (Eq4)
Initial: $\ce{4K + _O2 -> 2K2O}$; O: 2 on left, 2 on right → coefficient 1 for $\ce{O2}$
Step5: Balance H/O atoms (Eq5)
Initial: $\ce{2H2O2 -> _H2O + 2O2}$; Adjust: $\ce{2H2O2 -> 2H2O + 1O2}$ (H:4, O:4 on both sides)
Step6: Balance Cl atoms (Eq6)
Initial: $\ce{2Al + _Cl2 -> 2AlCl3}$; Cl: 6 on right → coefficient 3 for $\ce{Cl2}$
Step7: Balance Ag/O atoms (Eq7)
Initial: $\ce{2Ag2O -> _Ag + 4O2}$; Adjust: $\ce{2Ag2O -> 4Ag + 1O2}$ (Ag:4, O:2 on both sides)
Step8: Balance N atoms (Eq8)
Initial: $\ce{3H2 + _N2 -> 2NH3}$; N: 2 on left, 2 on right → coefficient 1 for $\ce{N2}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{1}$ $\ce{O2}$ (balanced: $\ce{2H2 + 1O2 -> 2H2O}$)
- $\boldsymbol{1}$ $\ce{O2}$ (balanced: $\ce{2Mg + 1O2 -> 2MgO}$)
- $\boldsymbol{1}$ $\ce{F2}$ (balanced: $\ce{2Li + 1F2 -> 2LiF}$)
- $\boldsymbol{1}$ $\ce{O2}$ (balanced: $\ce{4K + 1O2 -> 2K2O}$)
- $\boldsymbol{2}$ $\ce{H2O}$ (balanced: $\ce{2H2O2 -> 2H2O + 1O2}$)
- $\boldsymbol{3}$ $\ce{Cl2}$ (balanced: $\ce{2Al + 3Cl2 -> 2AlCl3}$)
- $\boldsymbol{4}$ $\ce{Ag}$ (balanced: $\ce{2Ag2O -> 4Ag + 1O2}$)
- $\boldsymbol{1}$ $\ce{N2}$ (balanced: $\ce{3H2 + 1N2 -> 2NH3}$)