QUESTION IMAGE
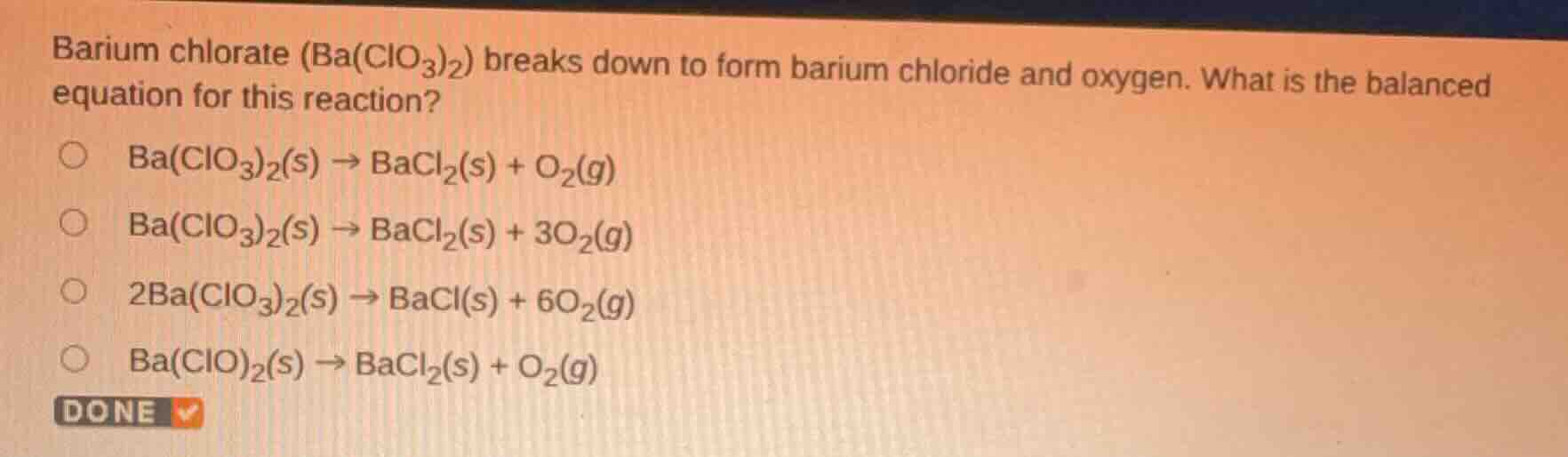
Question
barium chlorate (ba(clo₃)₂) breaks down to form barium chloride and oxygen. what is the balanced equation for this reaction?
ba(clo₃)₂(s) → bacl₂(s) + o₂(g)
ba(clo₃)₂(s) → bacl₂(s) + 3o₂(g)
2ba(clo₃)₂(s) → bacl(s) + 6o₂(g)
ba(clo)₂(s) → bacl₂(s) + o₂(g)
Step1: Analyze Ba atoms
On the left side, we have 1 Ba atom from $\ce{Ba(ClO_{3})_{2}}$. On the right side, in the correct product (barium chloride is $\ce{BaCl_{2}}$), we need 1 Ba atom. So Ba is balanced for now.
Step2: Analyze Cl atoms
Left side: 2 Cl atoms (from $\ce{Ba(ClO_{3})_{2}}$). Right side: In $\ce{BaCl_{2}}$, we have 2 Cl atoms. So Cl is balanced when we have $\ce{BaCl_{2}}$ as the product.
Step3: Analyze O atoms
Left side: In $\ce{Ba(ClO_{3})_{2}}$, there are $2\times3 = 6$ O atoms. Right side: In $\ce{O_{2}}$, let the coefficient of $\ce{O_{2}}$ be $x$. Then $2x=6$, so $x = 3$. So the balanced equation for O is 3 $\ce{O_{2}}$.
Putting it all together, the reactant is $\ce{Ba(ClO_{3})_{2}(s)}$, product is $\ce{BaCl_{2}(s)}$ and $3\ce{O_{2}(g)}$. So the balanced equation is $\ce{Ba(ClO_{3})_{2}(s)
ightarrow BaCl_{2}(s) + 3O_{2}(g)}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\ce{Ba(ClO_{3})_{2}(s)
ightarrow BaCl_{2}(s) + 3O_{2}(g)}$