QUESTION IMAGE
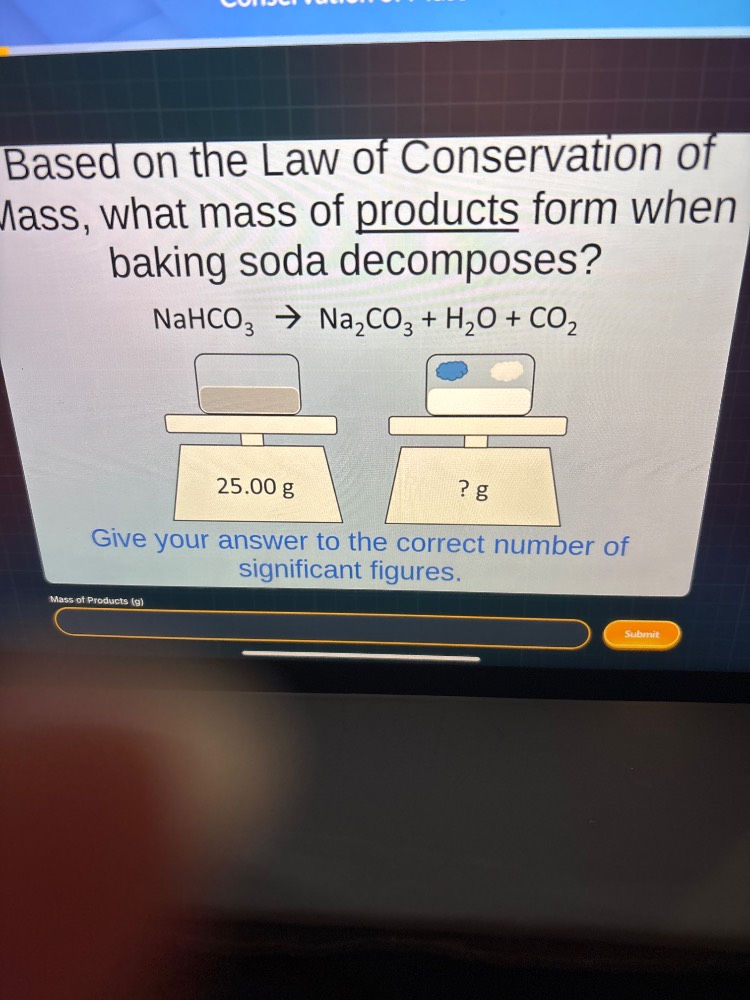
Question
based on the law of conservation of mass, what mass of products form when baking soda decomposes?
$\text{nahco}_3
ightarrow \text{na}_2\text{co}_3 + \text{h}_2\text{o} + \text{co}_2$
25.00 g
? g
give your answer to the correct number of significant figures.
mass of products (g)
Step1: Recall Law of Conservation of Mass
The Law of Conservation of Mass states that the total mass of reactants equals the total mass of products in a chemical reaction.
Step2: Apply the law to the reaction
The mass of the reactant (baking soda) is 25.00 g. No mass is lost or gained, so the total mass of products equals the mass of the reactant.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
25.00 g