QUESTION IMAGE
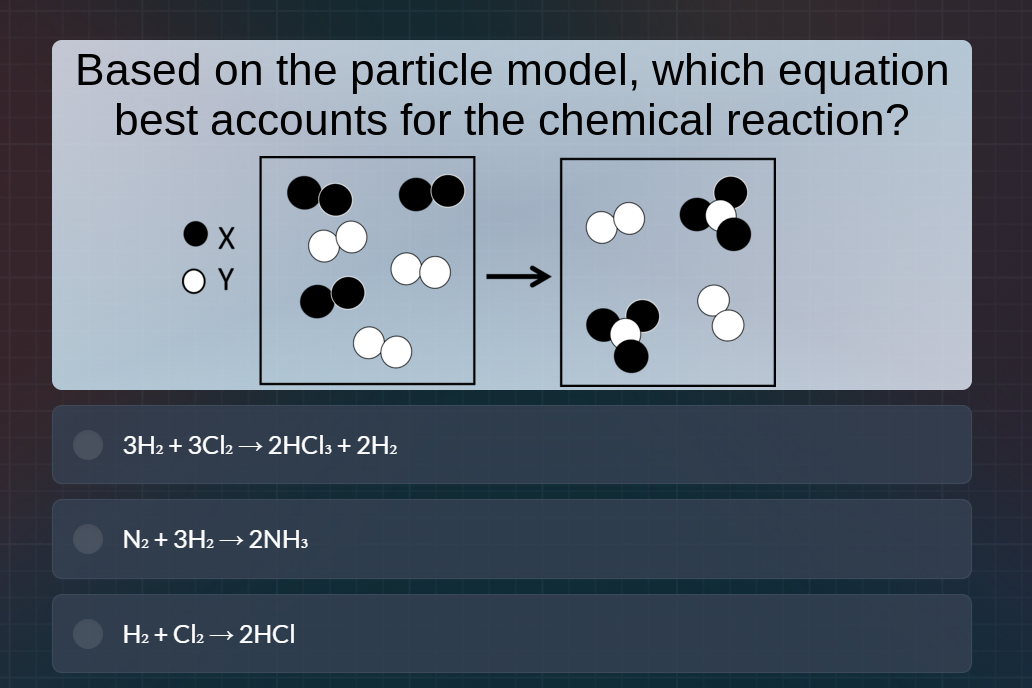
Question
based on the particle model, which equation best accounts for the chemical reaction?
x (black), y (white)
left box (reactants): three black diatomic (x₂), three white diatomic (y₂)
right box (products): two white diatomic (y₂), two molecules with one white and three black (y x₃)
options:
- 3h₂ + 3cl₂ → 2hcl₃ + 2h₂
- n₂ + 3h₂ → 2nh₃
- h₂ + cl₂ → 2hcl
- Analyze the particle model: Count the number of reactant molecules (X - X and Y - Y) and product molecules (X - Y - X? Wait, no, looking at the diagram: Left side has 3 black - black (X₂) and 3 white - white (Y₂)? Wait, no, let's re - examine. Wait, the left box: black - black (let's say X₂) molecules: 3 (since 3 pairs of black), white - white (Y₂) molecules: 3? Wait no, the left has 3 black - black (X₂) and 3 white - white (Y₂)? Wait no, the left: black - black: 3 (three groups of two black), white - white: 3 (three groups of two white)? Wait no, the right box: product has two molecules with three black and one white? No, wait the right box: two molecules with two black and one white? Wait no, the key is to match the reaction. Let's check the options:
- Option 1: \(3H_2 + 3Cl_2
ightarrow2HCl_3 + 2H_2\) is invalid as \(HCl_3\) is not a valid compound.
- Option 2: \(N_2+3H_2
ightarrow2NH_3\): Let's see the particle counts. If X is N (black) and Y is H (white). Left: 1 \(N_2\) (but in the diagram left has 3 black - black? Wait no, maybe I mis - counted. Wait the left box: black - black (X₂) : 3? No, wait the left has 3 black - black (X₂) and 3 white - white (Y₂)? No, the left has 3 black - black (X₂) and 3 white - white (Y₂)? Wait no, the left: black - black: 3 (three pairs), white - white: 3 (three pairs). The right: product has two molecules with three black and one white? No, wait the right has two molecules with two black and one white? No, the correct reaction for \(N_2 + 3H_2
ightarrow2NH_3\): 1 \(N_2\) (2 N atoms) and 3 \(H_2\) (6 H atoms) form 2 \(NH_3\) (2 N and 6 H). But in the diagram, left has 3 black - black (X₂) and 3 white - white (Y₂)? Wait no, maybe the diagram is for \(N_2+3H_2
ightarrow2NH_3\). Wait the first option has invalid product, the third option \(H_2 + Cl_2
ightarrow2HCl\) would have 1 \(H_2\) and 1 \(Cl_2\) forming 2 \(HCl\), but the diagram has more reactants. Wait no, maybe the diagram is: left has 1 \(N_2\) (black - black) and 3 \(H_2\) (white - white), forming 2 \(NH_3\) (each \(NH_3\) has 1 N and 3 H? No, \(NH_3\) has 1 N and 3 H, so 2 \(NH_3\) has 2 N and 6 H, which matches 1 \(N_2\) (2 N) and 3 \(H_2\) (6 H). The first option is invalid, the third option is a 1:1 reaction, but the diagram shows a 1:3 ratio (if X is N and Y is H). So the correct option is the second one.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \(N_2 + 3H_2
ightarrow2NH_3\)