QUESTION IMAGE
Question
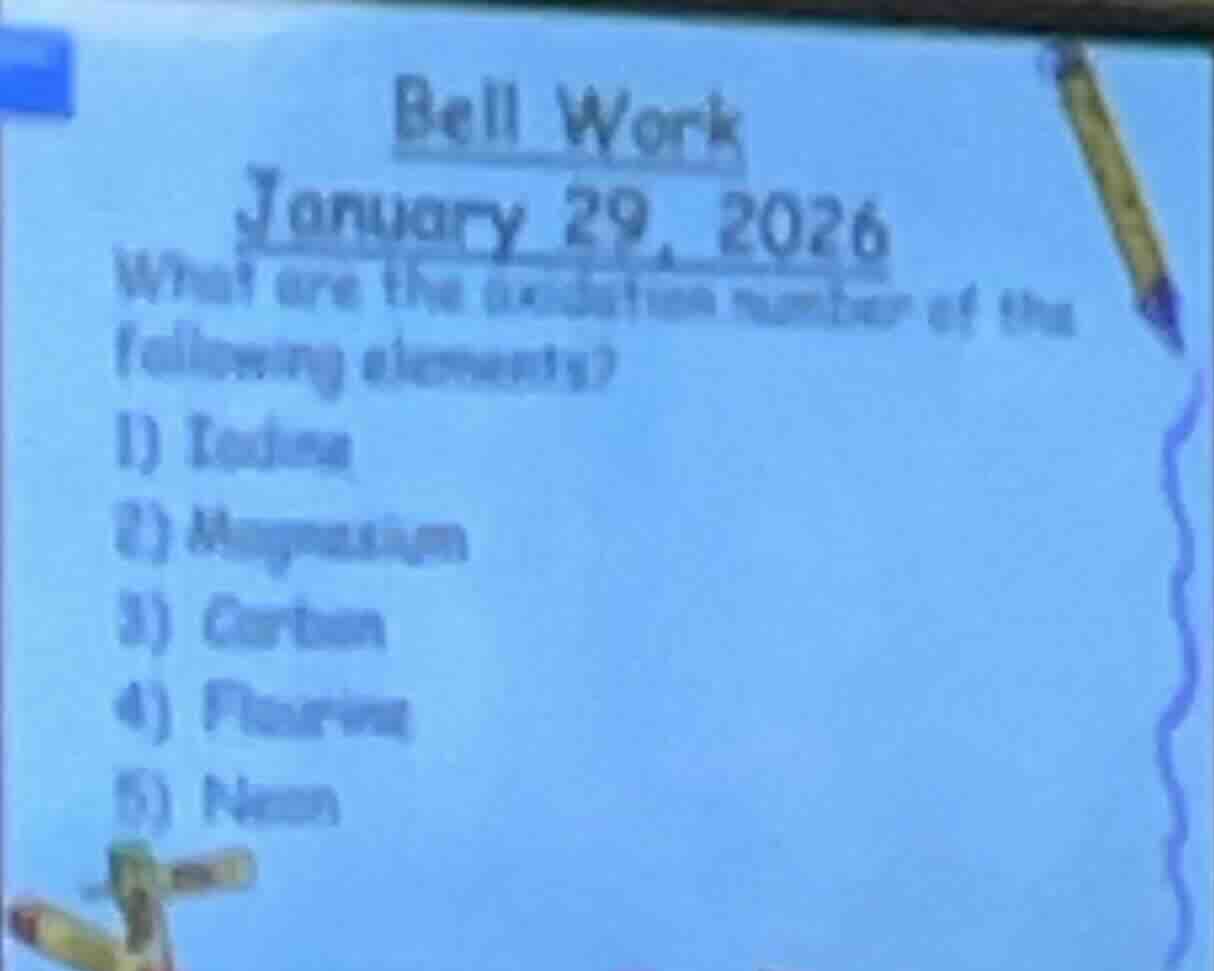
bell work
january 29, 2026
what are the oxidation number of the following elements?
- iodine
- magnesium
- carbon
- fluorine
- neon
To find the oxidation number of each element, we use the following rules:
- For a free element (an element in its elemental form, not combined with other elements), the oxidation number is 0.
- For a monatomic ion, the oxidation number is equal to the charge of the ion.
- In compounds, the sum of oxidation numbers of all atoms is equal to the overall charge of the compound (0 for neutral compounds, equal to the charge for ions).
Step 1: Iodine (I)
Iodine is a free element (assuming it's in its elemental form, like \(I_2\)). For a free element, the oxidation number is 0.
Step 2: Magnesium (Mg)
Magnesium is a free element (in its elemental form, \(Mg\)). The oxidation number of a free element is 0. If it's in a compound (like \(MgO\)), magnesium is a group 2 metal, so it has an oxidation number of +2 (since group 2 metals typically have a +2 oxidation state in compounds). But if we consider elemental magnesium, it's 0. Assuming the question is about elemental magnesium (since it's just "Magnesium" without a compound context), oxidation number is 0. If it's in a compound, we need more info, but likely elemental here, so 0.
Step 3: Carbon (C)
Carbon is a free element (elemental carbon, like graphite or \(C\)): oxidation number 0. In compounds, carbon can have various oxidation numbers (e.g., +4 in \(CO_2\), -4 in \(CH_4\), etc.). But without a compound context, assuming elemental carbon, oxidation number 0.
Step 4: Fluorine (F)
Fluorine is a free element (elemental fluorine, \(F_2\)): oxidation number 0. In compounds, fluorine is the most electronegative element, so it always has an oxidation number of -1. But if it's elemental \(F_2\), oxidation number is 0.
Step 5: Neon (Ne)
Neon is a noble gas, a free element (in its elemental form, \(Ne\)). The oxidation number of a free element is 0.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Iodine: 0 (if elemental); in compounds, depends on the compound, but likely 0 here.
- Magnesium: 0 (if elemental); in compounds, +2 (group 2 metal).
- Carbon: 0 (if elemental); in compounds, variable (e.g., +4, -4, etc.).
- Fluorine: 0 (if elemental); in compounds, -1.
- Neon: 0 (elemental, noble gas).
(Note: If the question assumes the elements are in their elemental forms, all have an oxidation number of 0. If they are in specific compounds, we need more details, but based on the question's phrasing "the oxidation number of the following elements" (without compound context), we assume elemental forms, so all have oxidation number 0.)