QUESTION IMAGE
Question
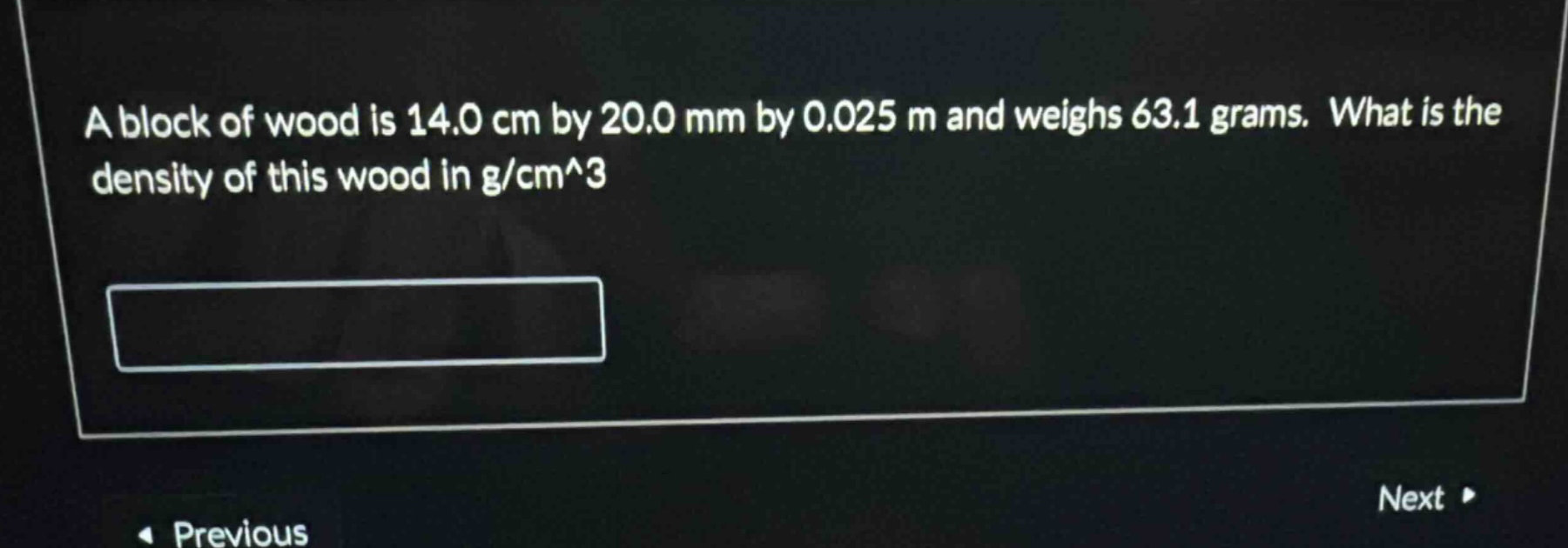
a block of wood is 14.0 cm by 20.0 mm by 0.025 m and weighs 63.1 grams. what is the density of this wood in g/cm^3
Step1: Convert all dimensions to cm
First, convert 20.0 mm to cm: since 1 cm = 10 mm, so \( 20.0\space mm=\frac{20.0}{10}\space cm = 2.00\space cm \).
Then, convert 0.025 m to cm: since 1 m = 100 cm, so \( 0.025\space m=0.025\times100\space cm = 2.5\space cm \).
The dimensions of the block are \( 14.0\space cm \), \( 2.00\space cm \), and \( 2.5\space cm \).
Step2: Calculate the volume of the block
The volume \( V \) of a rectangular block is given by \( V = l\times w\times h \).
Substituting the values: \( V=14.0\space cm\times2.00\space cm\times2.5\space cm \)
\( V = 14.0\times2.00\times2.5\space cm^3=70.0\space cm^3 \)
Step3: Calculate the density
Density \(
ho \) is given by the formula \(
ho=\frac{m}{V} \), where \( m = 63.1\space g \) and \( V = 70.0\space cm^3 \).
Substituting the values: \(
ho=\frac{63.1\space g}{70.0\space cm^3}\approx0.901\space g/cm^3 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.901\space g/cm^3 \) (or approximately \( 0.90\space g/cm^3 \) or \( 0.9\space g/cm^3 \) depending on significant figures, but the calculated value is approximately \( 0.901 \))