QUESTION IMAGE
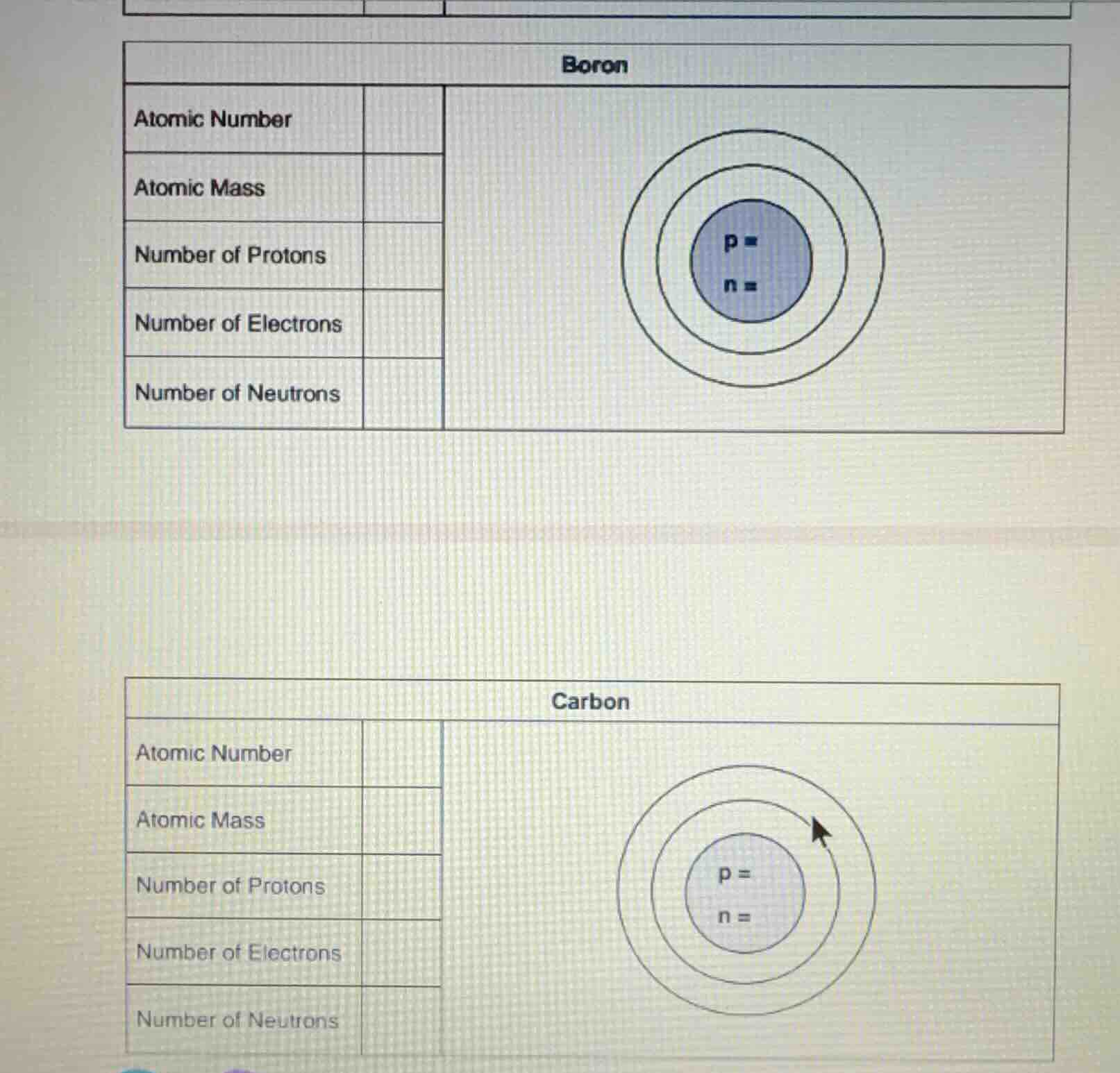
Question
boron
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
carbon
atomic number
atomic mass
number of protons
number of electrons
number of neutrons
p =
n =
Step1: Fill in Boron's core values
For boron (B):
- Atomic number = number of protons = number of electrons = 5
- Atomic mass (most common isotope, B-11) = 11
- Number of neutrons = atomic mass - atomic number = $11 - 5 = 6$
So, $p=5$, $n=6$ for boron's nucleus.
Step2: Fill in Carbon's core values
For carbon (C):
- Atomic number = number of protons = number of electrons = 6
- Atomic mass (most common isotope, C-12) = 12
- Number of neutrons = atomic mass - atomic number = $12 - 6 = 6$
So, $p=6$, $n=6$ for carbon's nucleus.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Boron Table:
| Category | Value |
|---|---|
| Atomic Mass | 11 |
| Number of Protons | 5 |
| Number of Electrons | 5 |
| Number of Neutrons | 6 |
Boron nucleus: $p=5$, $n=6$
Carbon Table:
| Category | Value |
|---|---|
| Atomic Mass | 12 |
| Number of Protons | 6 |
| Number of Electrons | 6 |
| Number of Neutrons | 6 |
Carbon nucleus: $p=6$, $n=6$