QUESTION IMAGE
Question
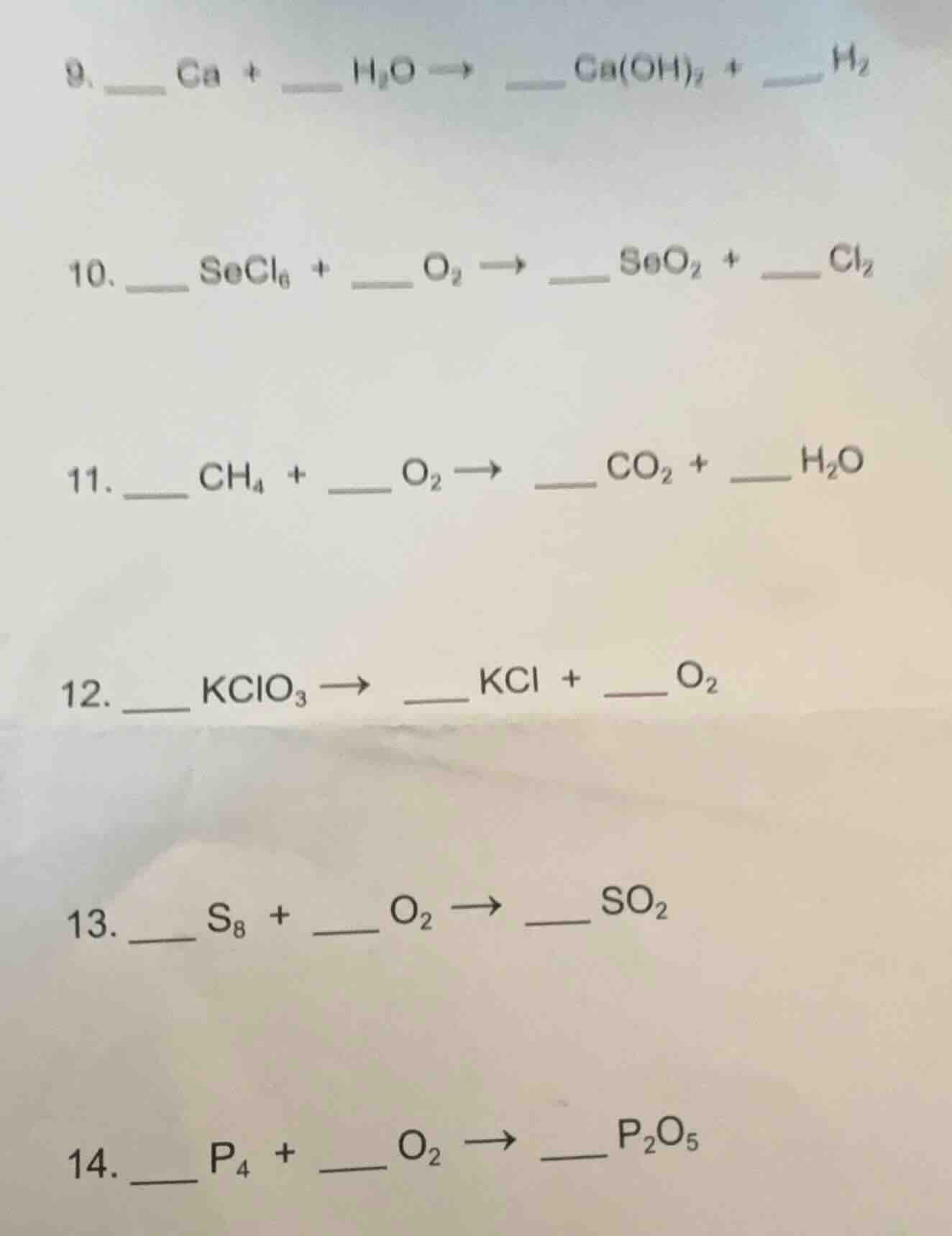
- _ ca + _ $h_2o$ → _ $ca(oh)_2$ + _ $h_2$
- _ $secl_6$ + _ $o_2$ → _ $seo_2$ + _ $cl_2$
- _ $ch_4$ + _ $o_2$ → _ $co_2$ + _ $h_2o$
- _ $kclo_3$ → _ $kcl$ + ___ $o_2$
- _ $s_8$ + _ $o_2$ → ___ $so_2$
- _ $p_4$ + _ $o_2$ → ___ $p_2o_5$
Step1: Balance Ca atoms
Ca: 1 on left, 1 on right. Coefficient for Ca = 1, $\text{Ca(OH)}_2$ = 1.
Step2: Balance H atoms
H: 2 on left, 4 on right. Coefficient for $\text{H}_2\text{O}$ = 2.
Step3: Balance O atoms
O: 2 on left, 2 on right. Balanced.
Step4: Verify all atoms
Ca:1=1, H:4=4, O:2=2. Equation 9 balanced.
---
Step1: Balance Se atoms
Se:1 on left,1 on right. Coefficient for $\text{SeCl}_6$=1, $\text{SeO}_2$=1.
Step2: Balance Cl atoms
Cl:6 on left,2 on right. Coefficient for $\text{Cl}_2$=3.
Step3: Balance O atoms
O:2 on left,2 on right. Coefficient for $\text{O}_2$=1.
Step4: Verify all atoms
Se:1=1, Cl:6=6, O:2=2. Equation10 balanced.
---
Step1: Balance C atoms
C:1 on left,1 on right. Coefficient for $\text{CH}_4$=1, $\text{CO}_2$=1.
Step2: Balance H atoms
H:4 on left,2 on right. Coefficient for $\text{H}_2\text{O}$=2.
Step3: Balance O atoms
O:2 on left,4 on right. Coefficient for $\text{O}_2$=2.
Step4: Verify all atoms
C:1=1, H:4=4, O:4=4. Equation11 balanced.
---
Step1: Balance K, Cl atoms
K,Cl:1 on left,1 on right. Coefficient for $\text{KClO}_3$=2, $\text{KCl}$=2.
Step2: Balance O atoms
O:6 on left,2 on right. Coefficient for $\text{O}_2$=3.
Step3: Verify all atoms
K:2=2, Cl:2=2, O:6=6. Equation12 balanced.
---
Step1: Balance S atoms
S:8 on left,1 on right. Coefficient for $\text{S}_8$=1, $\text{SO}_2$=8.
Step2: Balance O atoms
O:2 on left,16 on right. Coefficient for $\text{O}_2$=8.
Step3: Verify all atoms
S:8=8, O:16=16. Equation13 balanced.
---
Step1: Balance P atoms
P:4 on left,2 on right. Coefficient for $\text{P}_4$=1, $\text{P}_2\text{O}_5$=2.
Step2: Balance O atoms
O:2 on left,10 on right. Coefficient for $\text{O}_2$=5.
Step3: Verify all atoms
P:4=4, O:10=10. Equation14 balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{1\ \text{Ca} + 2\ \text{H}_2\text{O}
ightarrow 1\ \text{Ca(OH)}_2 + 1\ \text{H}_2}$
- $\boldsymbol{1\ \text{SeCl}_6 + 1\ \text{O}_2
ightarrow 1\ \text{SeO}_2 + 3\ \text{Cl}_2}$
- $\boldsymbol{1\ \text{CH}_4 + 2\ \text{O}_2
ightarrow 1\ \text{CO}_2 + 2\ \text{H}_2\text{O}}$
- $\boldsymbol{2\ \text{KClO}_3
ightarrow 2\ \text{KCl} + 3\ \text{O}_2}$
- $\boldsymbol{1\ \text{S}_8 + 8\ \text{O}_2
ightarrow 8\ \text{SO}_2}$
- $\boldsymbol{1\ \text{P}_4 + 5\ \text{O}_2
ightarrow 2\ \text{P}_2\text{O}_5}$