QUESTION IMAGE
Question
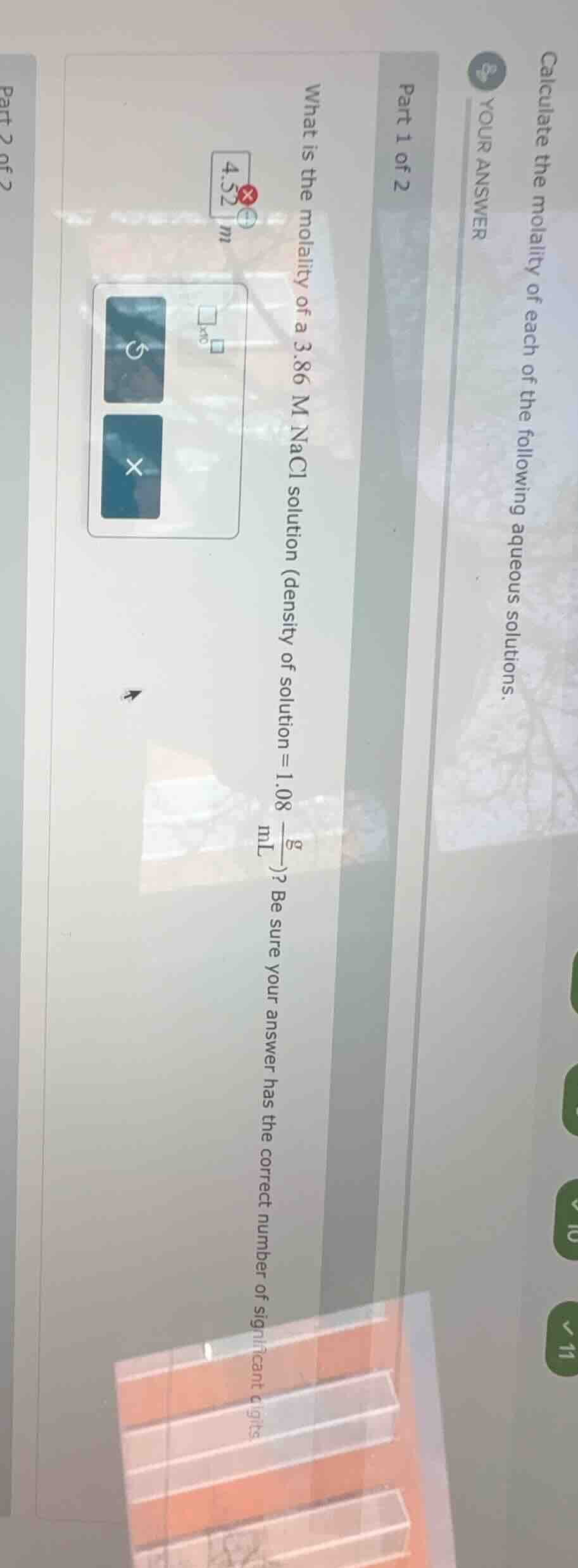
calculate the molality of each of the following aqueous solutions.
part 1 of 2
what is the molality of a 3.86 m nacl solution (density of solution = \\(\frac{1.08\\ g}{ml}\\))? be sure your answer has the correct number of significant digits.
Step1: Assume volume of solution
Let's assume the volume of the solution is \( V = 1.00\space L = 1000\space mL \).
Step2: Calculate moles of NaCl
Molarity \( M = \frac{n}{V} \), so moles of \( NaCl \), \( n = M\times V = 3.86\space mol/L\times1.00\space L = 3.86\space mol \).
Step3: Calculate mass of solution
Density \(
ho = \frac{m_{solution}}{V} \), so mass of solution \( m_{solution} =
ho\times V = 1.08\space g/mL\times1000\space mL = 1080\space g \).
Step4: Calculate mass of NaCl
Molar mass of \( NaCl = 58.44\space g/mol \), so mass of \( NaCl \), \( m_{NaCl} = n\times Molar\space mass = 3.86\space mol\times58.44\space g/mol \approx 225.6\space g \).
Step5: Calculate mass of solvent (water)
Mass of solvent \( m_{solvent} = m_{solution} - m_{NaCl} = 1080\space g - 225.6\space g = 854.4\space g = 0.8544\space kg \).
Step6: Calculate molality
Molality \( m = \frac{n}{m_{solvent}(in\space kg)} = \frac{3.86\space mol}{0.8544\space kg} \approx 4.52\space m \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 4.52\space m \)