QUESTION IMAGE
Question
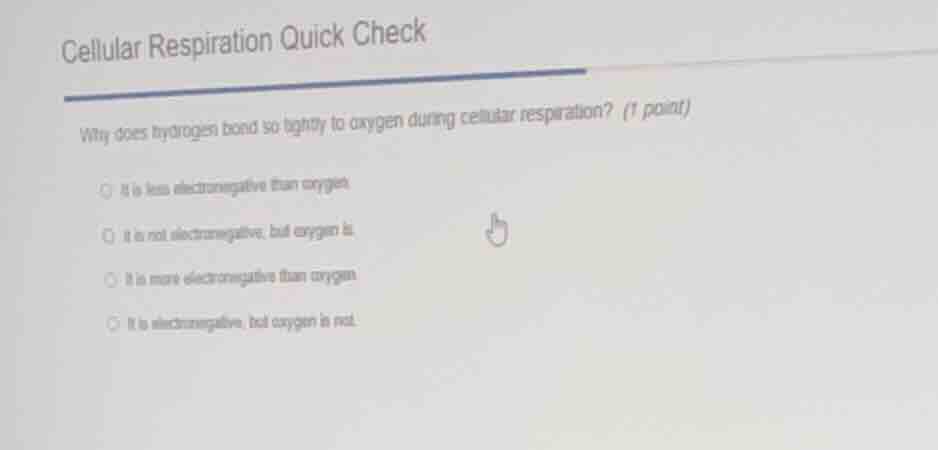
cellular respiration quick check
why does hydrogen bond so tightly to oxygen during cellular respiration? (1 point)
- it is less electronegative than oxygen
- it is not electronegative, but oxygen is
- it is more electronegative than oxygen
- it is electronegative, but oxygen is not
Brief Explanations
Electronegativity is the ability of an atom to attract shared electrons in a bond. Oxygen has a much higher electronegativity than hydrogen. This difference means oxygen pulls the shared electrons in the O-H bond closer to itself, creating a strong polar covalent bond between hydrogen and oxygen.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. It is less electronegative than oxygen