QUESTION IMAGE
Question
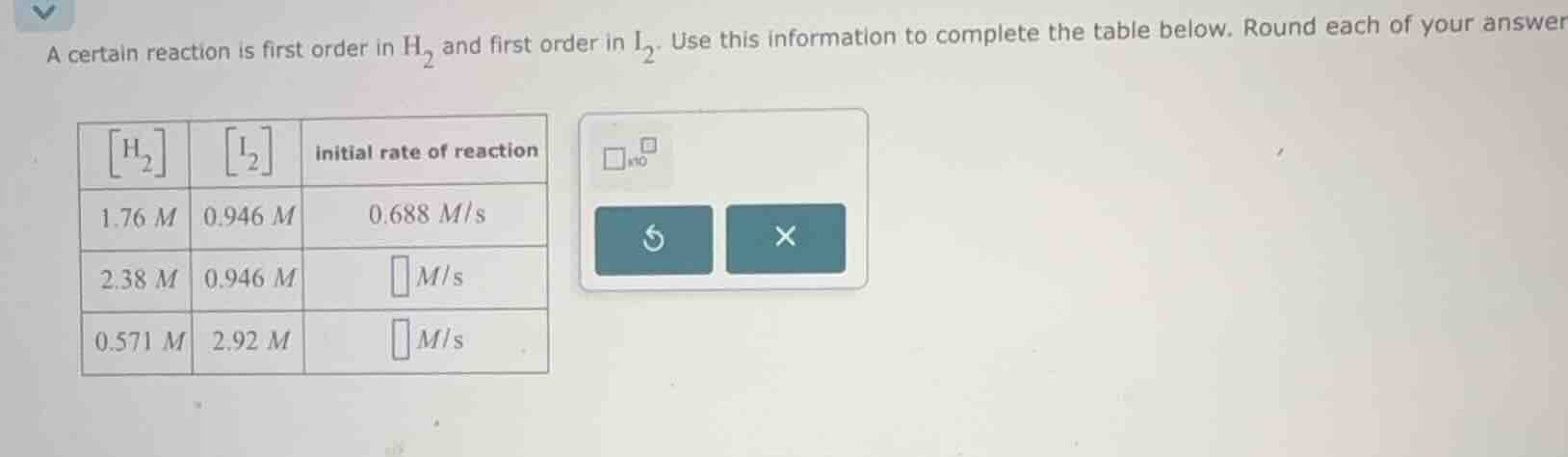
a certain reaction is first order in $ce{h_2}$ and first order in $ce{i_2}$. use this information to complete the table below. round each of your answer
| $ce{h_2}$ | $ce{i_2}$ | initial rate of reaction | ---- | ---- | ---- | 1.76 $m$ | 0.946 $m$ | 0.688 $m/s$ | 2.38 $m$ | 0.946 $m$ | $square$ $m/s$ | 0.571 $m$ | 2.92 $m$ | $square$ $m/s$ |
Step1: Write rate law equation
Since the reaction is first order in $\text{H}_2$ and first order in $\text{I}_2$, the rate law is:
$$\text{Rate} = k[\text{H}_2][\text{I}_2]$$
Step2: Solve for rate constant $k$
Use the first row of data to find $k$:
$$k = \frac{\text{Rate}}{[\text{H}_2][\text{I}_2]} = \frac{0.688\ \text{M/s}}{(1.76\ \text{M})(0.946\ \text{M})}$$
$$k = \frac{0.688}{1.66496} \approx 0.4132\ \text{M}^{-1}\text{s}^{-1}$$
Step3: Calculate rate for row 2
Substitute $k$, $[\text{H}_2]=2.38\ \text{M}$, $[\text{I}_2]=0.946\ \text{M}$ into rate law:
$$\text{Rate} = (0.4132)(2.38)(0.946)$$
$$\text{Rate} \approx 0.4132 \times 2.25148 \approx 0.930\ \text{M/s}$$
Step4: Calculate rate for row 3
Substitute $k$, $[\text{H}_2]=0.571\ \text{M}$, $[\text{I}_2]=2.92\ \text{M}$ into rate law:
$$\text{Rate} = (0.4132)(0.571)(2.92)$$
$$\text{Rate} \approx 0.4132 \times 1.66732 \approx 0.689\ \text{M/s}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Row 2 initial rate: $0.930\ \text{M/s}$
Row 3 initial rate: $0.689\ \text{M/s}$