QUESTION IMAGE
Question
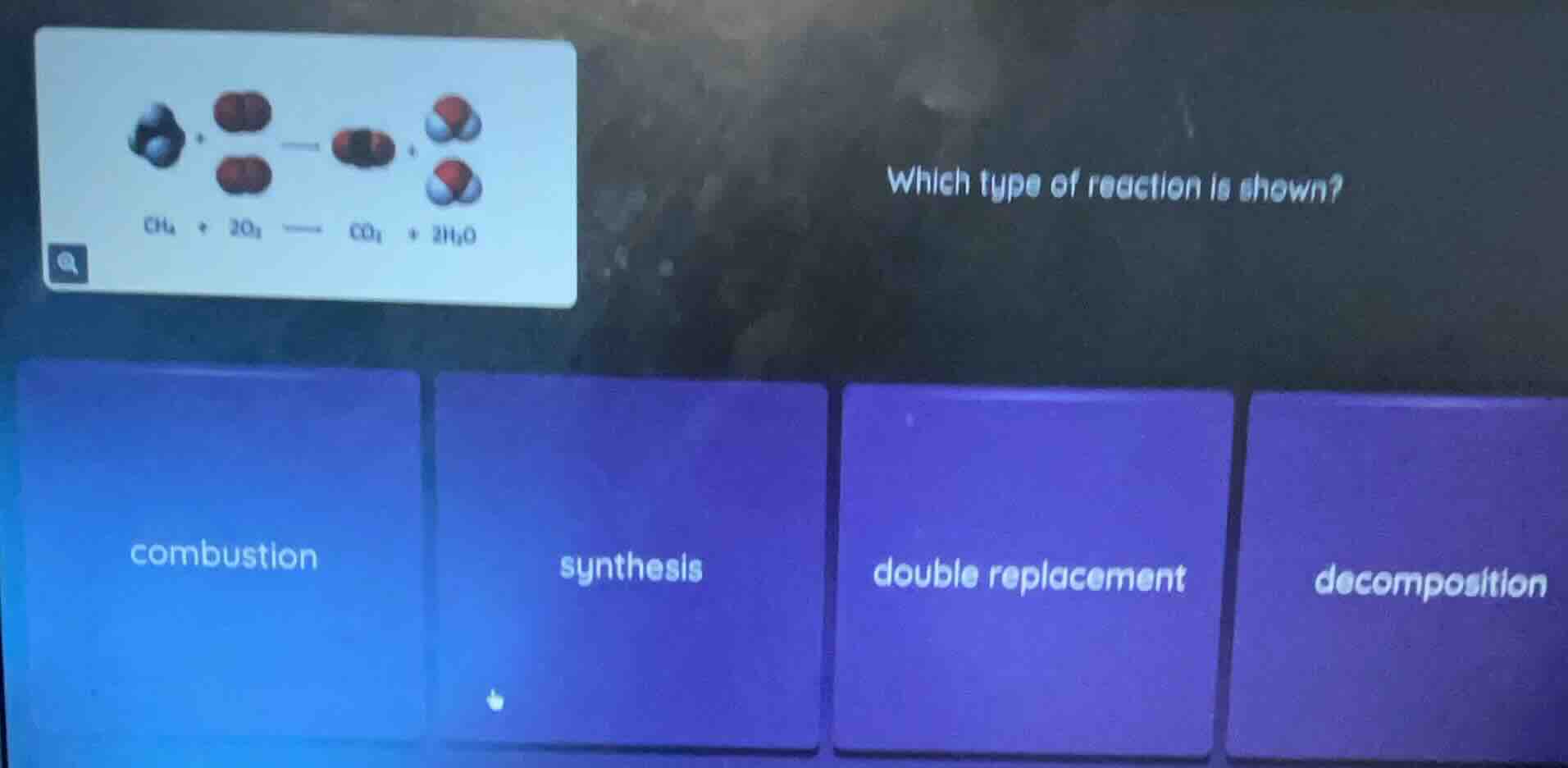
ch₄ + 2o₂ —— co₂ + 2h₂o which type of reaction is shown? combustion synthesis double replacement decomposition
The reaction is \( \text{CH}_4 + 2\text{O}_2
ightarrow \text{CO}_2 + 2\text{H}_2\text{O} \). A combustion reaction typically involves a hydrocarbon (like \( \text{CH}_4 \), methane) reacting with oxygen (\( \text{O}_2 \)) to produce carbon dioxide (\( \text{CO}_2 \)) and water (\( \text{H}_2\text{O} \)), along with the release of energy. Synthesis reactions combine substances into a single product, double - replacement involves swapping ions between two compounds, and decomposition breaks a compound into simpler substances. This reaction matches the pattern of combustion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. combustion