QUESTION IMAGE
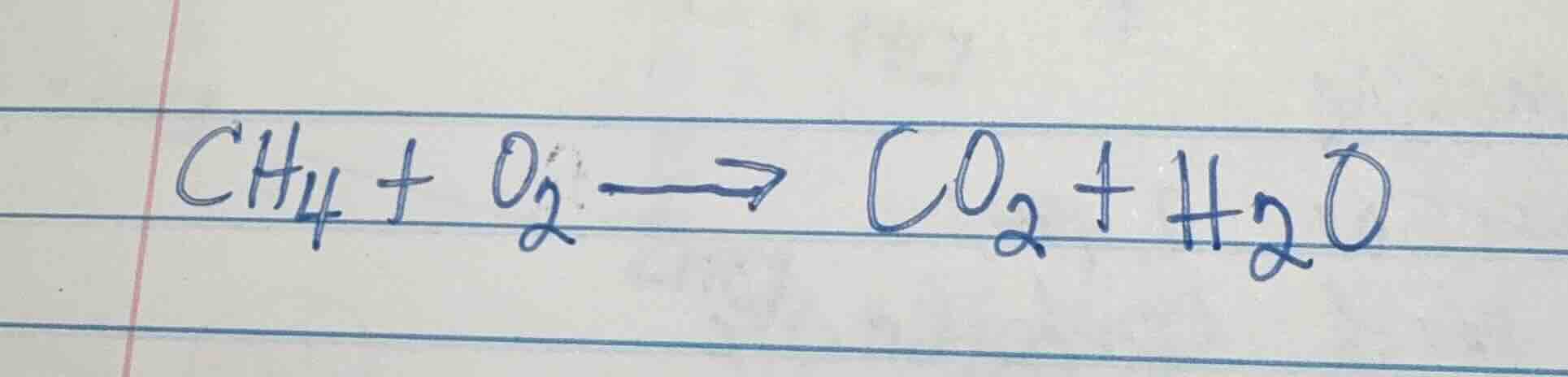
Question
ch₄ + o₂ → co₂ + h₂o
Step1: Balance Carbon atoms
On the left, we have 1 C in $CH_4$. On the right, we have 1 C in $CO_2$. So carbon is balanced for now.
Step2: Balance Hydrogen atoms
Left: 4 H in $CH_4$. Right: 2 H in $H_2O$. So we need to put a coefficient of 2 in front of $H_2O$ to make H atoms 4 (2×2). Now the equation is $CH_4 + O_2
ightarrow CO_2 + 2H_2O$.
Step3: Balance Oxygen atoms
Now, right side: $CO_2$ has 2 O, and $2H_2O$ has 2×1 = 2 O, total O on right is 2 + 2 = 4. Left side: $O_2$ has 2 O per molecule. So we need a coefficient of 2 in front of $O_2$ to get 4 O atoms. The balanced equation is $CH_4 + 2O_2
ightarrow CO_2 + 2H_2O$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced chemical equation is $\boldsymbol{CH_4 + 2O_2
ightarrow CO_2 + 2H_2O}$