QUESTION IMAGE
Question
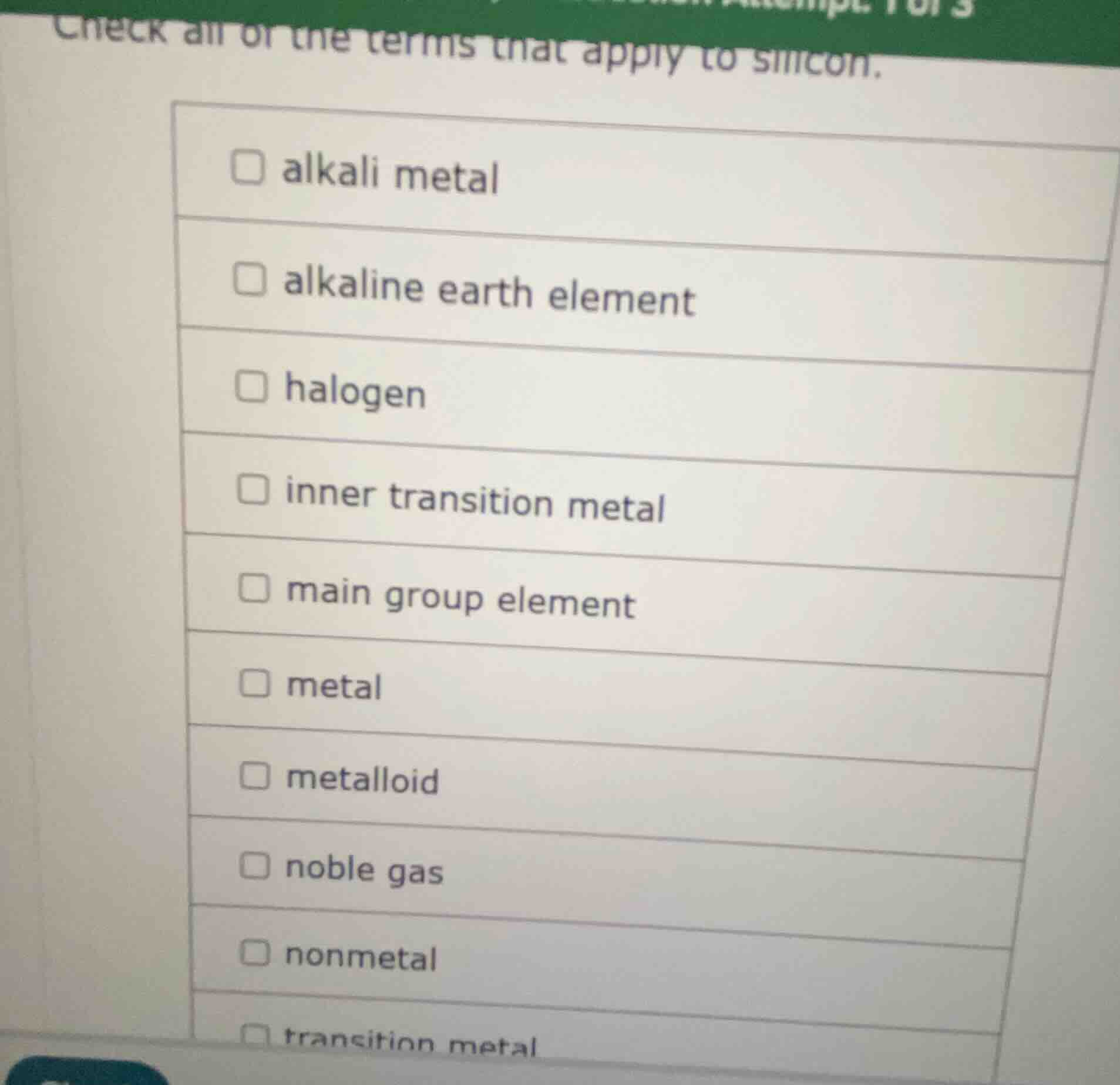
check all of the terms that apply to silicon.
□ alkali metal
□ alkaline earth element
□ halogen
□ inner transition metal
□ main group element
□ metal
□ metalloid
□ noble gas
□ nonmetal
□ transition metal
Brief Explanations
- Alkali metal: Group 1 elements (Li, Na, K, etc.). Silicon (Si) is in Group 14, so no.
- Alkaline earth element: Group 2 elements (Be, Mg, Ca, etc.). Si is Group 14, so no.
- Halogen: Group 17 elements (F, Cl, Br, etc.). Si is Group 14, so no.
- Inner transition metal: Lanthanides/actinides (f - block). Si is p - block, so no.
- Main group element: Elements in s - and p - blocks. Si is in p - block (Group 14), so yes.
- Metal: Si has properties of both metals and non - metals but is not a typical metal (e.g., poor electrical conductor compared to metals, brittle in solid form). So no.
- Metalloid: Elements with properties between metals and non - metals (B, Si, Ge, As, Sb, Te, Po). Si is a metalloid, so yes.
- Noble gas: Group 18 elements (He, Ne, Ar, etc.). Si is Group 14, so no.
- Nonmetal: Si has some non - metal properties but is classified as a metalloid. However, sometimes in broad terms, it can be considered a non - metal - like metalloid, but more accurately, metalloid. But among the options, main group element and metalloid apply. Also, silicon is a non - metal? Wait, no: metalloids are a separate category. But silicon is a metalloid and a main group element. Let's re - check:
- Main group elements: s and p block. Si is p block (Group 14), so main group element.
- Metalloid: Si is a metalloid (has properties of both metals and non - metals).
- Nonmetal: Silicon is often considered a metalloid, but in some classifications, it can be grouped with non - metals, but the key correct ones here are main group element and metalloid. Also, silicon is a non - metal? Wait, no, metalloid is a better fit. But let's confirm:
- Silicon is a metalloid (so the "metalloid" box should be checked).
- It is a main group element (so "main group element" box should be checked).
- Is it a nonmetal? Silicon has some non - metal properties (e.g., forms covalent bonds, is a semiconductor), but it is more accurately a metalloid. However, in the context of this question, the correct terms are main group element, metalloid, and nonmetal? Wait, no, let's check the periodic table:
- Silicon (Si) is in Group 14, Period 3.
- Main group elements: s and p block. Si is p block, so main group element: yes.
- Metalloid: yes (properties between metal and non - metal: semiconductor, brittle, forms covalent compounds, etc.).
- Nonmetal: Some sources consider metalloids as a subset of non - metals in a broad sense, but the strict classification has metalloids as a separate group. But in the options given, the terms that apply are main group element, metalloid, and nonmetal? Wait, no, let's check each option again:
- Alkali metal: No.
- Alkaline earth element: No.
- Halogen: No.
- Inner transition metal: No.
- Main group element: Yes (p - block, Group 14).
- Metal: No.
- Metalloid: Yes.
- Noble gas: No.
- Nonmetal: Silicon is a metalloid, but sometimes in basic classifications, it can be considered a non - metal. But the most accurate among the options are main group element and metalloid. Also, silicon is a non - metal? Wait, no, metalloid is a distinct category. But according to the standard classification, silicon is a metalloid and a main group element. So the terms that apply are main group element, metalloid, and nonmetal? Wait, no, let's see:
- Silicon is a metalloid (so "metalloid" is correct).
- It is a main group element (so "main group element" is correct).…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The terms that apply to silicon are:
- main group element
- metalloid
- nonmetal
(To mark the boxes: check "main group element", "metalloid", and "nonmetal")