QUESTION IMAGE
Question
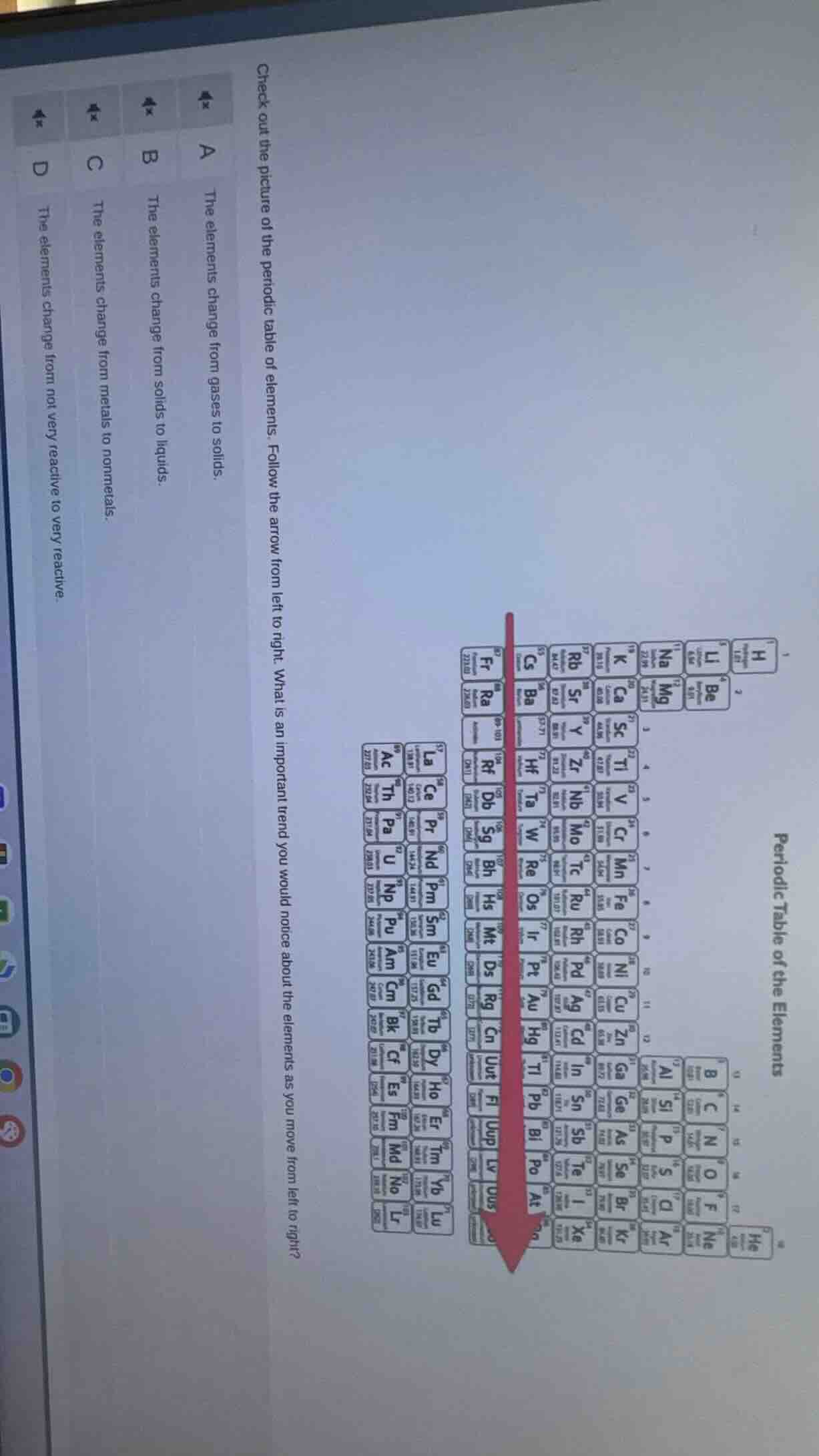
check out the picture of the periodic table of elements. follow the arrow from left to right. what is an important trend you would notice about the elements as you move from left to right?
a the elements change from gases to solids.
b the elements change from solids to liquids.
c the elements change from metals to nonmetals.
d the elements change from not very reactive to very reactive.
Brief Explanations
To solve this, we analyze each option based on the periodic table's left - to - right trends:
- Option A: The elements do not follow a gas - to - solid trend from left to right. For example, on the left, we have metals (solids like Na), and on the right, we have non - metals which can be gases (like O₂, N₂) or solids (like S). So this is incorrect.
- Option B: The general state - of - matter trend from left to right is not a simple solid - to - liquid change. Most elements on the left are solids (metals), and on the right, we have a mix of solids, liquids (like Br₂), and gases. So this is incorrect.
- Option C: As we move from left to right across a period in the periodic table, the elements change from metals (on the left, e.g., Na, Mg) to non - metals (on the right, e.g., O, F). This is a well - known periodic trend.
- Option D: The reactivity trend for metals (left side) decreases from left to right, and for non - metals (right side) increases from left to right. So the overall trend is not from not very reactive to very reactive. This is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The elements change from metals to nonmetals.