QUESTION IMAGE
Question
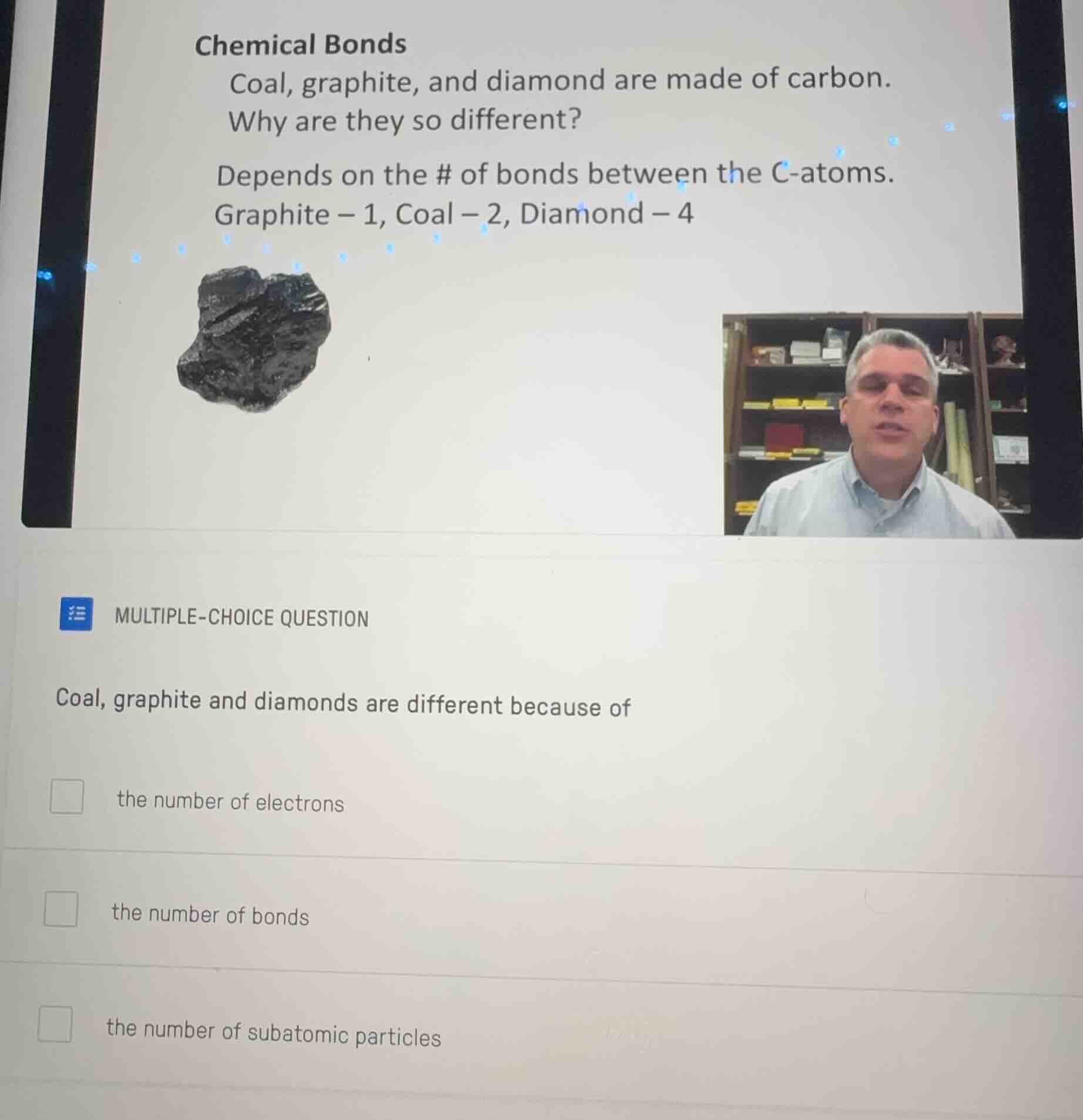
chemical bonds
coal, graphite, and diamond are made of carbon.
why are they so different?
depends on the # of bonds between the c - atoms.
graphite - 1, coal - 2, diamond - 4
multiple - choice question
coal, graphite and diamonds are different because of
the number of electrons
the number of bonds
the number of subatomic particles
The text states that coal, graphite, and diamond (all carbon - based) differ because of the number of bonds between carbon atoms. The option "the number of electrons" is incorrect as these substances are all carbon (same number of electrons in carbon atoms). The option "the number of subatomic particles" is incorrect as they are all carbon (same subatomic particle composition for carbon atoms). Only "the number of bonds" matches the given explanation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
the number of bonds (the option with the text "the number of bonds")