QUESTION IMAGE
Question
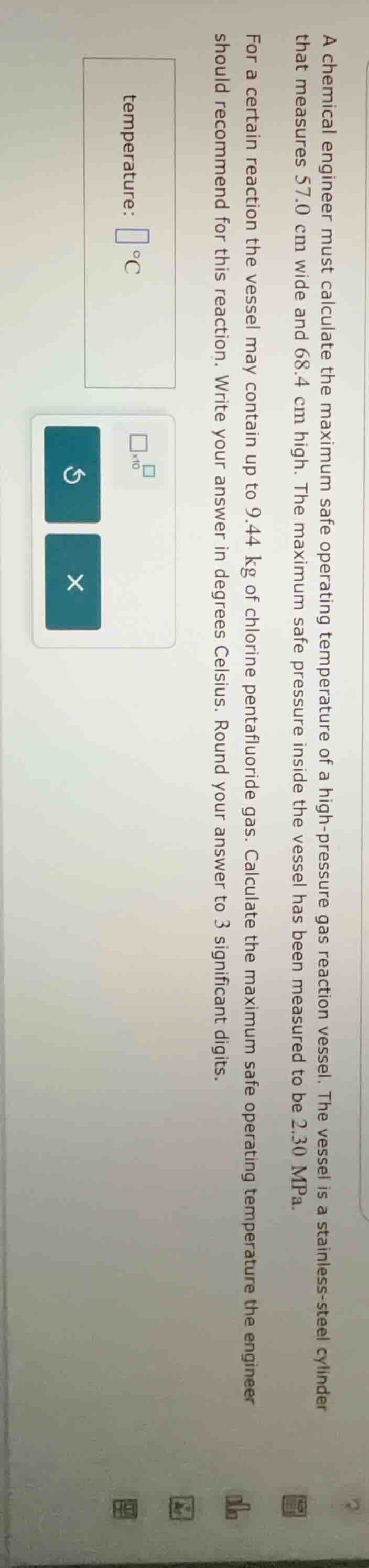
a chemical engineer must calculate the maximum safe operating temperature of a high - pressure gas reaction vessel. the vessel is a stainless - steel cylinder that measures 57.0 cm wide and 68.4 cm high. the maximum safe pressure inside the vessel has been measured to be 2.30 mpa. for a certain reaction the vessel may contain up to 9.44 kg of chlorine pentafluoride gas. calculate the maximum safe operating temperature the engineer should recommend for this reaction. write your answer in degrees celsius. round your answer to 3 significant digits.
Step 1: Determine the volume of the cylinder
The vessel is a cylinder with diameter \(d = 57.0\space cm\), so radius \(r=\frac{d}{2}=\frac{57.0}{2} = 28.5\space cm\) and height \(h = 68.4\space cm\). The volume of a cylinder is given by \(V=\pi r^{2}h\).
\[
\]
Step 2: Calculate the moles of chlorine pentafluoride (\(ClF_{5}\))
The molar mass of \(ClF_{5}\): \(Cl = 35.45\space g/mol\), \(F = 19.00\space g/mol\), so molar mass \(M=35.45 + 5\times19.00=35.45 + 95.00 = 130.45\space g/mol\).
Mass \(m = 9.44\space kg=9440\space g\). Moles \(n=\frac{m}{M}=\frac{9440}{130.45}\approx72.37\space mol\).
Step 3: Use the ideal gas law \(PV = nRT\) to find temperature \(T\)
Pressure \(P = 2.30\space MPa=2.30\times10^{6}\space Pa = 2.30\times10^{6}\space N/m^{2}\), volume \(V = 174.5\space L=0.1745\space m^{3}\), \(n = 72.37\space mol\), \(R = 8.314\space J/(mol\cdot K)\).
From \(PV=nRT\), \(T=\frac{PV}{nR}\).
\[
\]
Step 4: Convert temperature from Kelvin to Celsius
\(T(^{\circ}C)=T(K)-273.15\)
\[
T(^{\circ}C)=669.2 - 273.15 = 396.05\space^{\circ}C\approx396\space^{\circ}C
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(396^{\circ}C\)