QUESTION IMAGE
Question
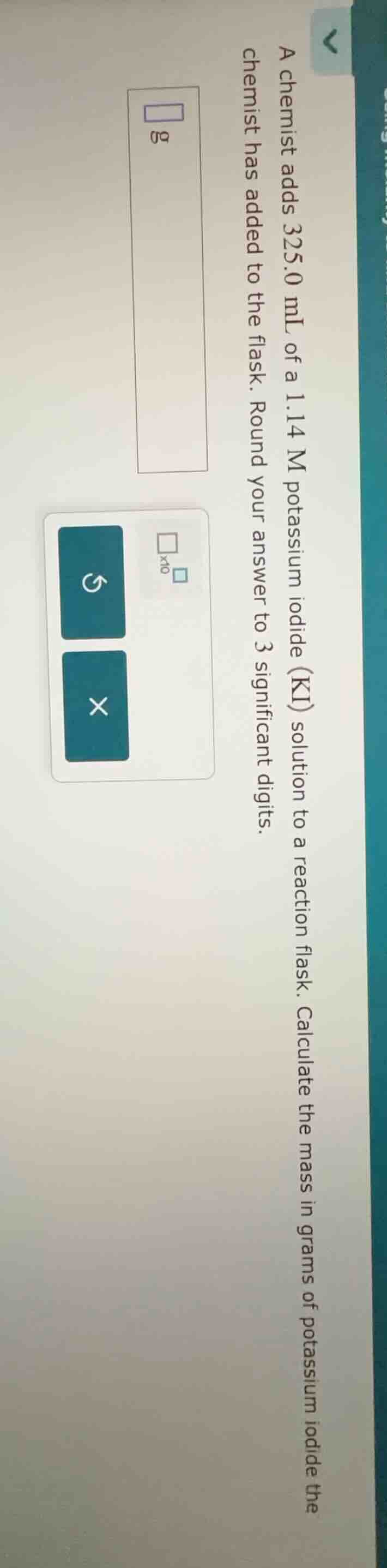
a chemist adds 325.0 ml of a 1.14 m potassium iodide (ki) solution to a reaction flask. calculate the mass in grams of potassium iodide the chemist has added to the flask. round your answer to 3 significant digits.
Step1: Convert volume to liters
$325.0\ \text{mL} = 325.0 \times 10^{-3}\ \text{L} = 0.3250\ \text{L}$
Step2: Calculate moles of KI
$\text{Moles of KI} = \text{Molarity} \times \text{Volume (L)} = 1.14\ \text{mol/L} \times 0.3250\ \text{L} = 0.3705\ \text{mol}$
Step3: Find molar mass of KI
Molar mass of K = $39.10\ \text{g/mol}$, molar mass of I = $126.90\ \text{g/mol}$
$\text{Molar mass of KI} = 39.10 + 126.90 = 166.00\ \text{g/mol}$
Step4: Calculate mass of KI
$\text{Mass} = \text{Moles} \times \text{Molar mass} = 0.3705\ \text{mol} \times 166.00\ \text{g/mol} = 61.503\ \text{g}$
Step5: Round to 3 significant digits
$61.503\ \text{g} \approx 61.5\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
61.5 g