QUESTION IMAGE
Question
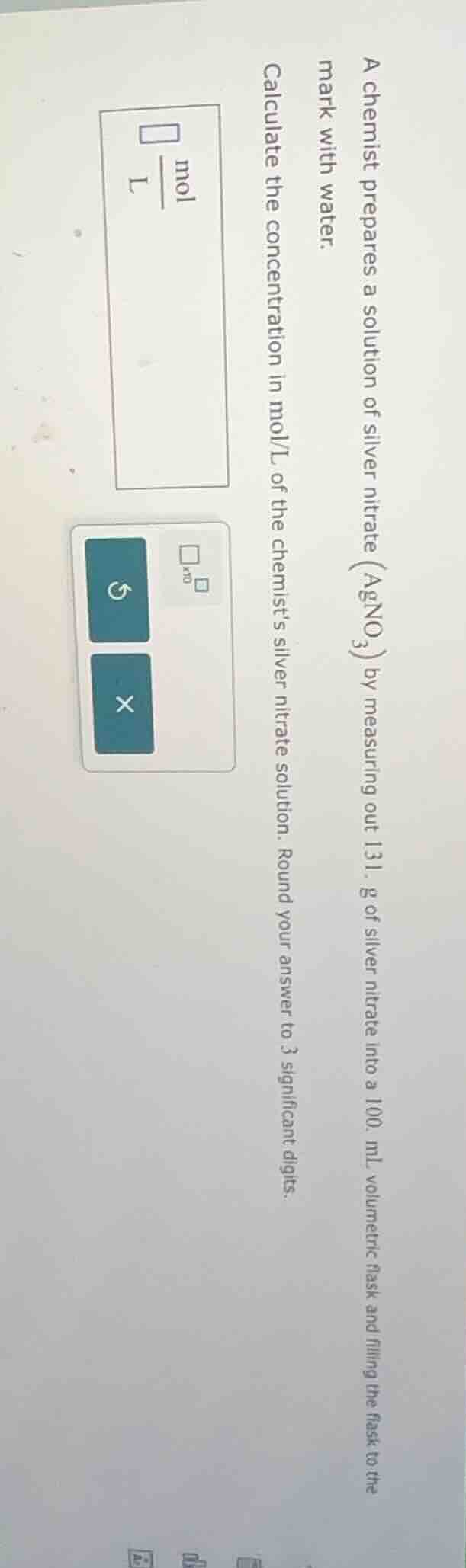
a chemist prepares a solution of silver nitrate ($\ce{agno_3}$) by measuring out 131. g of silver nitrate into a 100. ml volumetric flask and filling the flask to the mark with water. calculate the concentration in mol/l of the chemist’s silver nitrate solution. round your answer to 3 significant digits.
Step1: Calculate moles of \( AgNO_3 \)
Molar mass of \( AgNO_3 \): \( Ag = 107.87 \, g/mol \), \( N = 14.01 \, g/mol \), \( O = 16.00 \, g/mol \).
Molar mass \( M = 107.87 + 14.01 + 3\times16.00 = 169.88 \, g/mol \).
Moles \( n = \frac{mass}{molar \, mass} = \frac{131. \, g}{169.88 \, g/mol} \approx 0.771 \, mol \).
Step2: Convert volume to liters
Volume \( V = 100. \, mL = 0.100 \, L \) (since \( 1 \, L = 1000 \, mL \)).
Step3: Calculate molarity
Molarity \( M = \frac{n}{V} = \frac{0.771 \, mol}{0.100 \, L} = 7.71 \, mol/L \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 7.71 \)