QUESTION IMAGE
Question
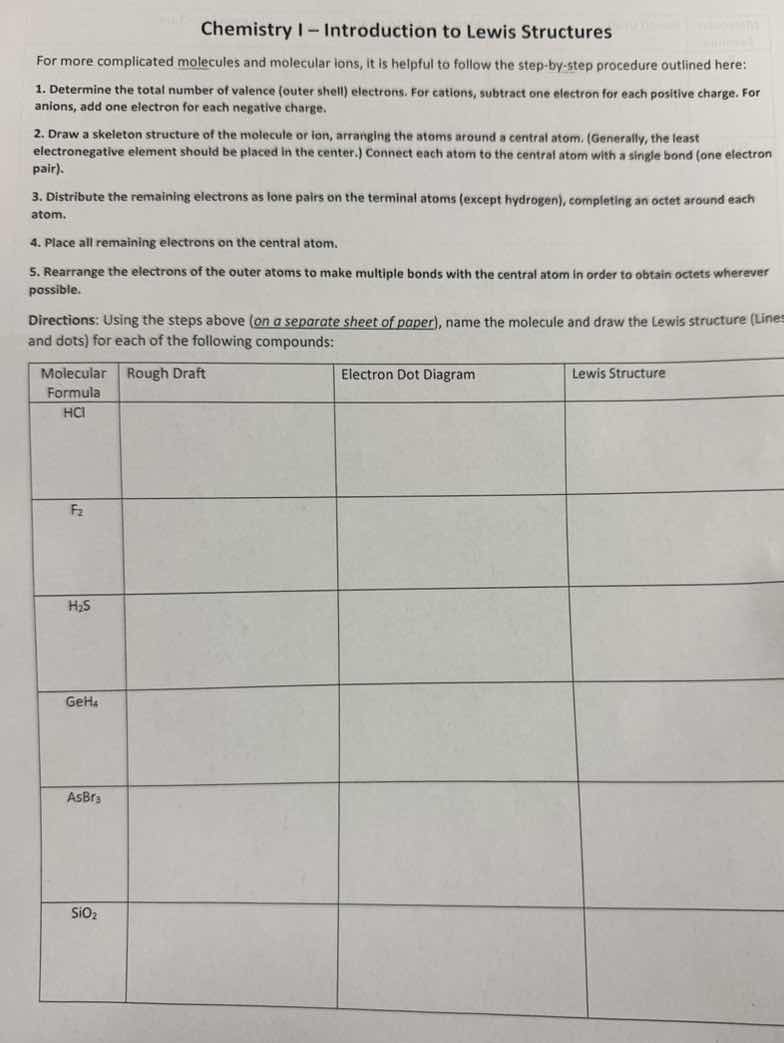
chemistry i – introduction to lewis structures
for more complicated molecules and molecular ions, it is helpful to follow the step - by - step procedure outlined here:
- determine the total number of valence (outer shell) electrons. for cations, subtract one electron for each positive charge. for anions, add one electron for each negative charge.
- draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom. (generally, the least electronegative element should be placed in the center.) connect each atom to the central atom with a single bond (one electron pair).
- distribute the remaining electrons as lone pairs on the terminal atoms (except hydrogen), completing an octet around each atom.
- place all remaining electrons on the central atom.
- rearrange the electrons of the outer atoms to make multiple bonds with the central atom in order to obtain octets wherever possible.
directions: using the steps above (on a separate sheet of paper), name the molecule and draw the lewis structure (lines and dots) for each of the following compounds:
| molecular formula | rough draft | electron dot diagram | lewis structure |
|---|---|---|---|
| f₂ | |||
| h₂s | |||
| geh₄ | |||
| asbr₃ | |||
| sio₂ |
To solve for the Lewis structures of each compound, we follow the 5 - step procedure for Lewis structures:
1. For \( \boldsymbol{HCl} \)
- Step 1: Determine total valence electrons
- Hydrogen (\( H \)) has 1 valence electron, Chlorine (\( Cl \)) has 7 valence electrons.
- Total valence electrons \( = 1 + 7=8 \).
- Step 2: Draw skeleton structure
- The less electronegative atom ( \( H \)) is bonded to \( Cl \) with a single bond: \( H - Cl \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the single bond (2 electrons), we have \( 8 - 2 = 6 \) electrons left. These are placed as lone pairs on \( Cl \) to complete its octet: \( H-\ddot{\underset{\cdot\cdot}{Cl}} \).
- Step 4: Place remaining electrons on central atom
- There are no remaining electrons to place on the central atom ( \( Cl \) already has an octet from the lone pairs and the bond).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- \( HCl \) has a single bond and \( Cl \) has an octet, so no multiple bonds are required.
2. For \( \boldsymbol{F_2} \)
- Step 1: Determine total valence electrons
- Each Fluorine (\( F \)) atom has 7 valence electrons. For \( F_2 \), total valence electrons \( = 7\times2 = 14 \).
- Step 2: Draw skeleton structure
- The two \( F \) atoms are bonded with a single bond: \( F - F \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the single bond (2 electrons), we have \( 14 - 2=12 \) electrons left. These are distributed as lone pairs on each \( F \) atom. Each \( F \) atom gets 3 lone pairs (6 electrons) to complete their octets: \( \ddot{\underset{\cdot\cdot}{F}}-\ddot{\underset{\cdot\cdot}{F}} \).
- Step 4: Place remaining electrons on central atom
- There are no remaining electrons to place on the central atom (both \( F \) atoms have octets).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octets are already satisfied with a single bond and lone pairs.
3. For \( \boldsymbol{H_2S} \)
- Step 1: Determine total valence electrons
- Hydrogen (\( H \)) has 1 valence electron (2 atoms, so \( 2\times1 = 2 \) valence electrons), Sulfur (\( S \)) has 6 valence electrons.
- Total valence electrons \( = 2 + 6=8 \).
- Step 2: Draw skeleton structure
- Sulfur (\( S \)) is the central atom (less electronegative than \( H \) in terms of bonding for this molecule) and is bonded to two \( H \) atoms: \( H - S - H \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the two single bonds (4 electrons), we have \( 8 - 4 = 4 \) electrons left. These are placed as two lone pairs on \( S \) to complete its octet: \( H-\ddot{S}-H \).
- Step 4: Place remaining electrons on central atom
- The remaining electrons (the two lone pairs) are already placed on the central atom (\( S \)).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octet of \( S \) is satisfied with the two single bonds and two lone pairs.
4. For \( \boldsymbol{GeH_4} \)
- Step 1: Determine total valence electrons
- Germanium (\( Ge \)) has 4 valence electrons, Hydrogen (\( H \)) has 1 valence electron (4 atoms, so \( 4\times1 = 4 \) valence electrons).
- Total valence electrons \( = 4+4 = 8 \).
- Step 2: Draw skeleton structure
- Germanium (\( Ge \)) is the central atom and is bonded to four \( H \) atoms: \( H - Ge - H \) (with two more \( H \) atoms bonded to \( Ge \) in a…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the Lewis structures of each compound, we follow the 5 - step procedure for Lewis structures:
1. For \( \boldsymbol{HCl} \)
- Step 1: Determine total valence electrons
- Hydrogen (\( H \)) has 1 valence electron, Chlorine (\( Cl \)) has 7 valence electrons.
- Total valence electrons \( = 1 + 7=8 \).
- Step 2: Draw skeleton structure
- The less electronegative atom ( \( H \)) is bonded to \( Cl \) with a single bond: \( H - Cl \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the single bond (2 electrons), we have \( 8 - 2 = 6 \) electrons left. These are placed as lone pairs on \( Cl \) to complete its octet: \( H-\ddot{\underset{\cdot\cdot}{Cl}} \).
- Step 4: Place remaining electrons on central atom
- There are no remaining electrons to place on the central atom ( \( Cl \) already has an octet from the lone pairs and the bond).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- \( HCl \) has a single bond and \( Cl \) has an octet, so no multiple bonds are required.
2. For \( \boldsymbol{F_2} \)
- Step 1: Determine total valence electrons
- Each Fluorine (\( F \)) atom has 7 valence electrons. For \( F_2 \), total valence electrons \( = 7\times2 = 14 \).
- Step 2: Draw skeleton structure
- The two \( F \) atoms are bonded with a single bond: \( F - F \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the single bond (2 electrons), we have \( 14 - 2=12 \) electrons left. These are distributed as lone pairs on each \( F \) atom. Each \( F \) atom gets 3 lone pairs (6 electrons) to complete their octets: \( \ddot{\underset{\cdot\cdot}{F}}-\ddot{\underset{\cdot\cdot}{F}} \).
- Step 4: Place remaining electrons on central atom
- There are no remaining electrons to place on the central atom (both \( F \) atoms have octets).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octets are already satisfied with a single bond and lone pairs.
3. For \( \boldsymbol{H_2S} \)
- Step 1: Determine total valence electrons
- Hydrogen (\( H \)) has 1 valence electron (2 atoms, so \( 2\times1 = 2 \) valence electrons), Sulfur (\( S \)) has 6 valence electrons.
- Total valence electrons \( = 2 + 6=8 \).
- Step 2: Draw skeleton structure
- Sulfur (\( S \)) is the central atom (less electronegative than \( H \) in terms of bonding for this molecule) and is bonded to two \( H \) atoms: \( H - S - H \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the two single bonds (4 electrons), we have \( 8 - 4 = 4 \) electrons left. These are placed as two lone pairs on \( S \) to complete its octet: \( H-\ddot{S}-H \).
- Step 4: Place remaining electrons on central atom
- The remaining electrons (the two lone pairs) are already placed on the central atom (\( S \)).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octet of \( S \) is satisfied with the two single bonds and two lone pairs.
4. For \( \boldsymbol{GeH_4} \)
- Step 1: Determine total valence electrons
- Germanium (\( Ge \)) has 4 valence electrons, Hydrogen (\( H \)) has 1 valence electron (4 atoms, so \( 4\times1 = 4 \) valence electrons).
- Total valence electrons \( = 4+4 = 8 \).
- Step 2: Draw skeleton structure
- Germanium (\( Ge \)) is the central atom and is bonded to four \( H \) atoms: \( H - Ge - H \) (with two more \( H \) atoms bonded to \( Ge \) in a tetrahedral arrangement, but for Lewis structure, we can represent it as \( H_3Ge - H \) or simply show the four single bonds).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the four single bonds (8 electrons), there are no remaining electrons to distribute as lone pairs on the terminal (\( H \)) atoms ( \( H \) only needs 2 electrons for a duet, which is satisfied by the single bond).
- Step 4: Place remaining electrons on central atom
- There are no remaining electrons to place on the central atom (\( Ge \) has a complete octet from the four single bonds).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octet of \( Ge \) is satisfied with the four single bonds.
5. For \( \boldsymbol{AsBr_3} \)
- Step 1: Determine total valence electrons
- Arsenic (\( As \)) has 5 valence electrons, Bromine (\( Br \)) has 7 valence electrons (3 atoms, so \( 3\times7 = 21 \) valence electrons).
- Total valence electrons \( = 5+21 = 26 \).
- Step 2: Draw skeleton structure
- Arsenic (\( As \)) is the central atom and is bonded to three \( Br \) atoms with single bonds: \( Br - As - Br \) (and one more \( Br \) bonded to \( As \)).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the three single bonds (6 electrons), we have \( 26 - 6=20 \) electrons left. Each \( Br \) atom needs 6 more electrons to complete its octet. For 3 \( Br \) atoms, we use \( 3\times6 = 18 \) electrons.
- After placing lone pairs on \( Br \) atoms: \( \ddot{\underset{\cdot\cdot}{Br}}-As-\ddot{\underset{\cdot\cdot}{Br}} \) (with the third \( Br \) also having 6 lone pair electrons: \( \ddot{\underset{\cdot\cdot}{Br}}-As-\ddot{\underset{\cdot\cdot}{Br}}-\ddot{\underset{\cdot\cdot}{Br}} \)).
- Step 4: Place remaining electrons on central atom
- We have used \( 6 + 18=24 \) electrons, so we have \( 26 - 24 = 2 \) electrons left. These are placed as a lone pair on \( As \): \( \ddot{\underset{\cdot\cdot}{Br}}-\underset{\cdot\cdot}{\ddot{As}}-\ddot{\underset{\cdot\cdot}{Br}}-\ddot{\underset{\cdot\cdot}{Br}} \).
- Step 5: Rearrange electrons for multiple bonds (not needed here)
- The octet of \( As \) (5 + 3 (bonds) + 2 (lone pair) = 10, which is an expanded octet, but this is acceptable for elements in the third period and below) and the octets of \( Br \) are satisfied.
6. For \( \boldsymbol{SiO_2} \)
- Step 1: Determine total valence electrons
- Silicon (\( Si \)) has 4 valence electrons, Oxygen (\( O \)) has 6 valence electrons (2 atoms, so \( 2\times6 = 12 \) valence electrons).
- Total valence electrons \( = 4 + 12=16 \).
- Step 2: Draw skeleton structure
- Silicon (\( Si \)) is the central atom and is bonded to two \( O \) atoms: \( O - Si - O \).
- Step 3: Distribute remaining electrons as lone pairs on terminal atoms
- After the two single bonds (4 electrons), we have \( 16 - 4 = 12 \) electrons left. We start by placing 6 electrons (3 lone pairs) on each \( O \) atom. But if we do this, \( Si \) will only have 4 electrons (from the two single bonds), which is less than an octet.
- Step 4: Place remaining electrons on central atom
- Since \( Si \) does not have an octet, we need to form multiple bonds.
- Step 5: Rearrange electrons for multiple bonds
- We convert one lone pair from each \( O \) atom into a double bond with \( Si \). The Lewis structure becomes \( O = Si = O \), with each \( O \) atom having two remaining lone pairs: \( \ddot{O}=Si=\ddot{O} \). In this structure, \( Si \) has an octet (4 electrons from the two double bonds) and each \( O \) has an octet (4 electrons from the double bond and 4 electrons from the two lone pairs).
Naming the Molecules
- \( HCl \): Hydrochloric acid (or Hydrogen chloride)
- \( F_2 \): Fluorine
- \( H_2S \): Hydrogen sulfide
- \( GeH_4 \): Germanium hydride (or Germane)
- \( AsBr_3 \): Arsenic tribromide
- \( SiO_2 \): Silicon dioxide
Lewis Structures Summary
| Molecular Formula | Rough Draft (Skeleton + Lone Pairs) | Electron Dot Diagram (Lone Pairs as Dots, Bonds as Lines) | Lewis Structure (Formal) |
|---|---|---|---|
| \( F_2 \) | \( \ddot{\underset{\cdot\cdot}{F}}-\ddot{\underset{\cdot\cdot}{F}} \) | \( :\ddot{\underset{\cdot\cdot}{F}}-\ddot{\underset{\cdot\cdot}{F}}: \) | \( :\ddot{F}-\ddot{F}: \) |
| \( H_2S \) | \( H-\ddot{S}-H \) | \( H:\ddot{S}:H \) | \( H - \ddot{S}-H \) |
| \( GeH_4 \) | \( H_3Ge - H \) (with 4 single bonds) | \( H:H:Ge:H:H \) (simplified, showing bonds as electron pairs) | \( H_3Ge - H \) (tetrahedral with 4 single bonds) |
| \( AsBr_3 \) | \( \ddot{\underset{\cdot\cdot}{Br}}-\underset{\cdot\cdot}{\ddot{As}}-\ddot{\underset{\cdot\cdot}{Br}}-\ddot{\underset{\cdot\cdot}{Br}} \) | \( :\ddot{Br}:\underset{\cdot\cdot}{\ddot{As}}:\ddot{Br}: :\ddot{Br}: \) (bonds as electron pairs between atoms) | \( \ddot{Br}-As-\ddot{Br}-\ddot{Br} \) (with lone pairs on \( As \) and \( Br \)) |
| \( SiO_2 \) | \( \ddot{O}=Si=\ddot{O} \) | \( :\ddot{O}=Si=\ddot{O}: \) | \( O = Si = O \) (with lone pairs on \( O \)) |
The key steps for each compound involve calculating valence electrons, drawing the skeleton, distributing lone pairs, and forming multiple bonds when necessary to satisfy the octet rule.