QUESTION IMAGE
Question
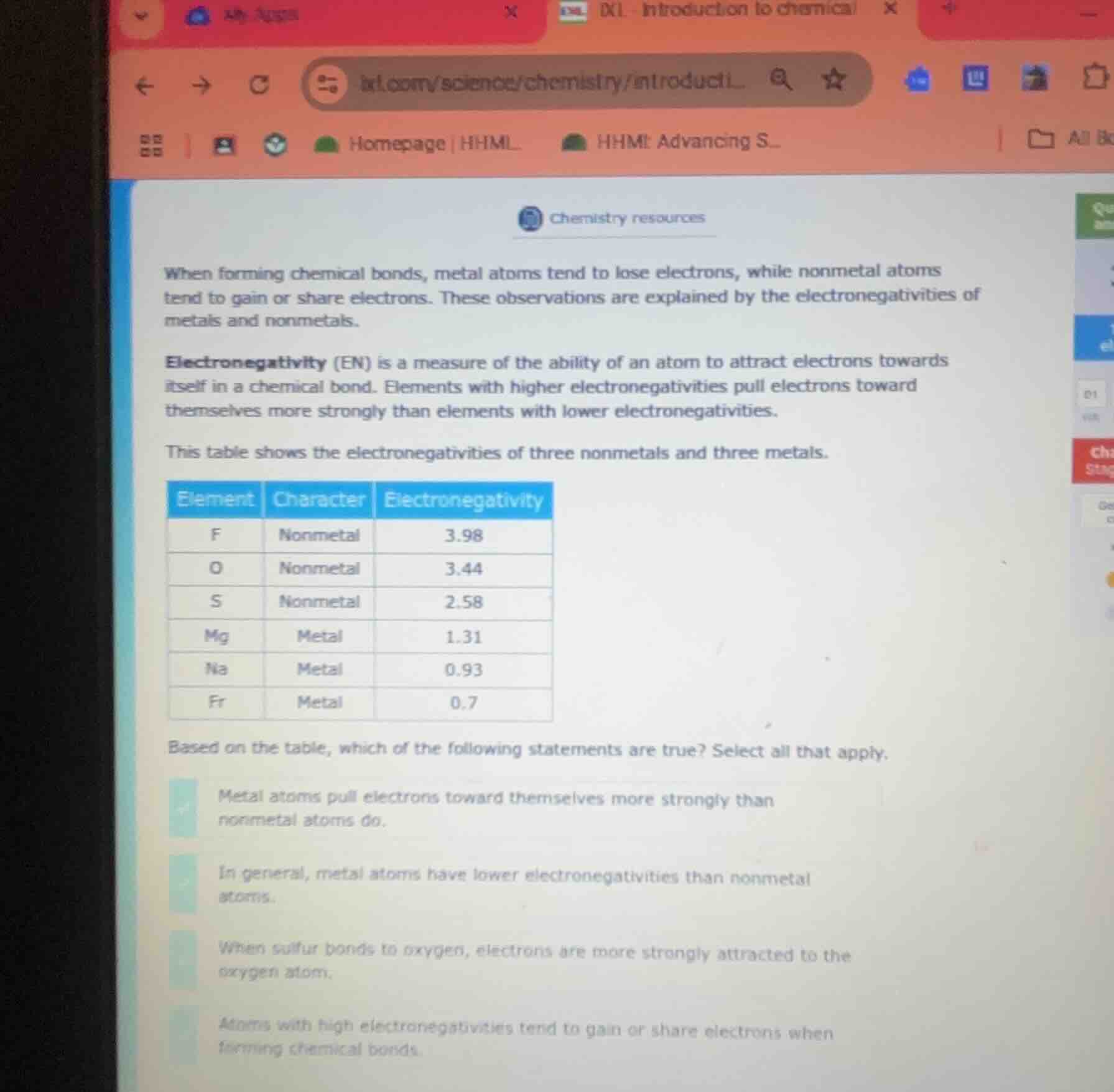
chemistry resources
when forming chemical bonds, metal atoms tend to lose electrons, while nonmetal atoms tend to gain or share electrons. these observations are explained by the electronegativities of metals and nonmetals.
electronegativity (en) is a measure of the ability of an atom to attract electrons towards itself in a chemical bond. elements with higher electronegativities pull electrons toward themselves more strongly than elements with lower electronegativities.
this table shows the electronegativities of three nonmetals and three metals.
| element | character | electronegativity | ---- | ---- | ---- | f | nonmetal | 3.98 | o | nonmetal | 3.44 | s | nonmetal | 2.58 | mg | metal | 1.31 | na | metal | 0.93 | fr | metal | 0.7 | based on the table, which of the following statements are true? select all that apply. |
- metal atoms pull electrons toward themselves more strongly than nonmetal atoms do.
- in general, metal atoms have lower electronegativities than nonmetal atoms.
- when sulfur bonds to oxygen, electrons are more strongly attracted to the oxygen atom.
- atoms with high electronegativities tend to gain or share electrons when forming chemical bonds.
Brief Explanations
- Compare the electronegativity values of metals and nonmetals in the table: all nonmetals have higher EN than metals, so metals do not pull electrons stronger.
- From the table, nonmetals (F:3.98, O:3.44, S:2.58) have higher EN than metals (Mg:1.31, Na:0.93, Fr:0.7), so this general statement holds.
- Oxygen has an electronegativity of 3.44, sulfur has 2.58. Higher EN means stronger electron attraction, so electrons are pulled more to oxygen.
- The text states nonmetals (high EN) gain/share electrons, which matches this statement.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- In general, metal atoms have lower electronegativities than nonmetal atoms.
- When sulfur bonds to oxygen, electrons are more strongly attracted to the oxygen atom.
- Atoms with high electronegativities tend to gain or share electrons when forming chemical bonds.