QUESTION IMAGE
Question
- choose the best answer.
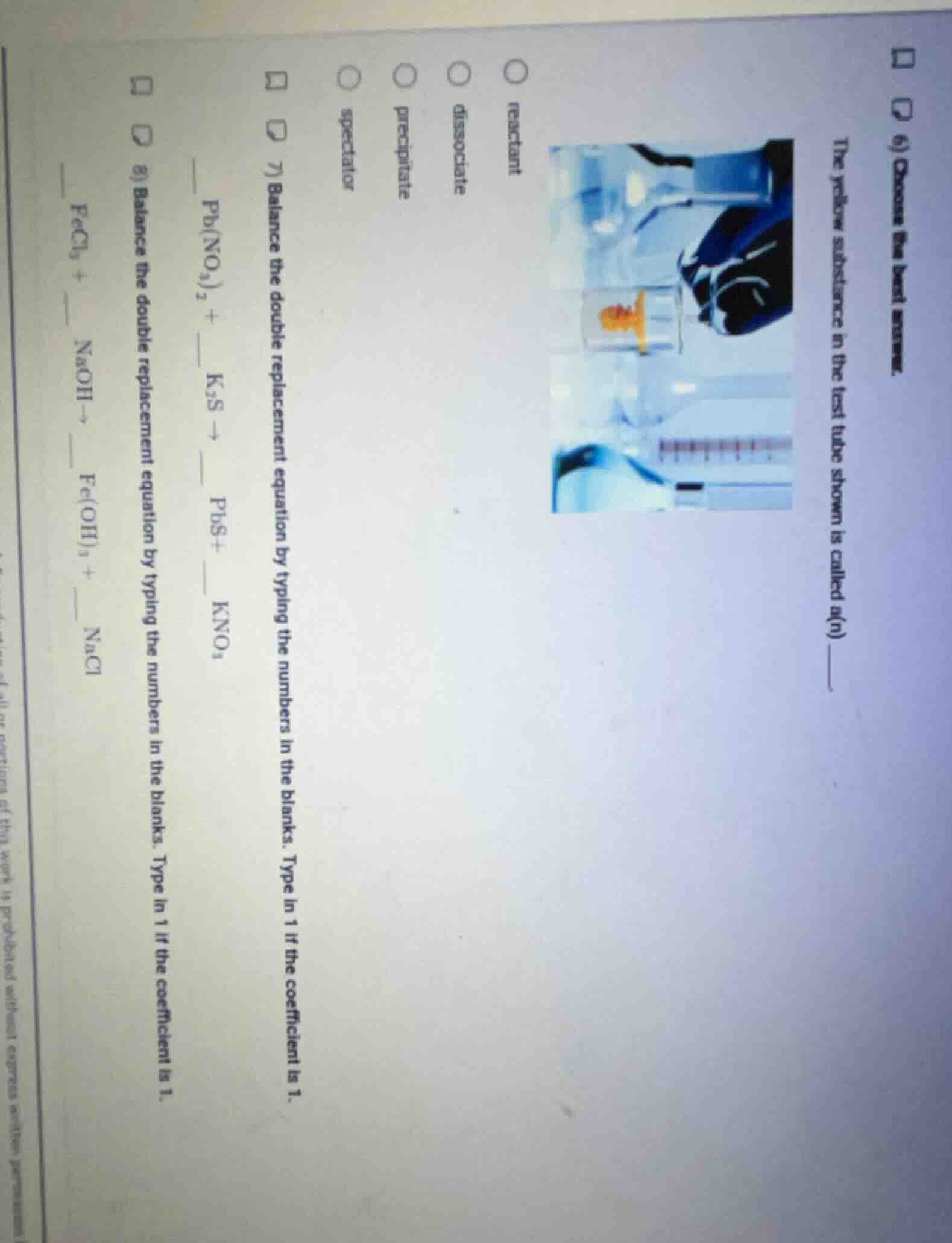
the yellow substance in the test tube shown is called a(n) ______
options: reactant, dissociate, precipitate, spectator
- balance the double replacement equation by typing the numbers in the blanks. type in 1 if the coefficient is 1.
__ pb(no₃)₂ + k₂s → pbs + __ kno₃
- balance the double replacement equation by typing the numbers in the blanks. type in 1 if the coefficient is 1.
__ fecl₃ + naoh → fe(oh)₃ + __ nacl
(Question 6):
Step1: Identify the substance type
The yellow solid forms as an insoluble product from mixing solutions, which is a precipitate.
(Question 7):
Step1: Balance Pb atoms
Pb is balanced (1 on each side).
Step2: Balance S atoms
S is balanced (1 on each side).
Step3: Balance K and NO₃⁻
There are 2 K and 2 NO₃⁻ on left, so add 2 to KNO₃.
$$\ce{Pb(NO3)2 + K2S -> PbS + 2KNO3}$$
Step4: Verify all atoms
Pb:1, N:2, O:6, K:2, S:1 on both sides.
(Question 8):
Step1: Balance Fe atoms
Fe is balanced (1 on each side).
Step2: Balance Cl atoms
3 Cl on left, add 3 to NaCl.
$$\ce{FeCl3 + NaOH -> Fe(OH)3 + 3NaCl}$$
Step3: Balance Na atoms
3 Na on right, add 3 to NaOH.
$$\ce{FeCl3 + 3NaOH -> Fe(OH)3 + 3NaCl}$$
Step4: Verify OH⁻ atoms
3 OH⁻ on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Question 6):
precipitate
---