QUESTION IMAGE
Question
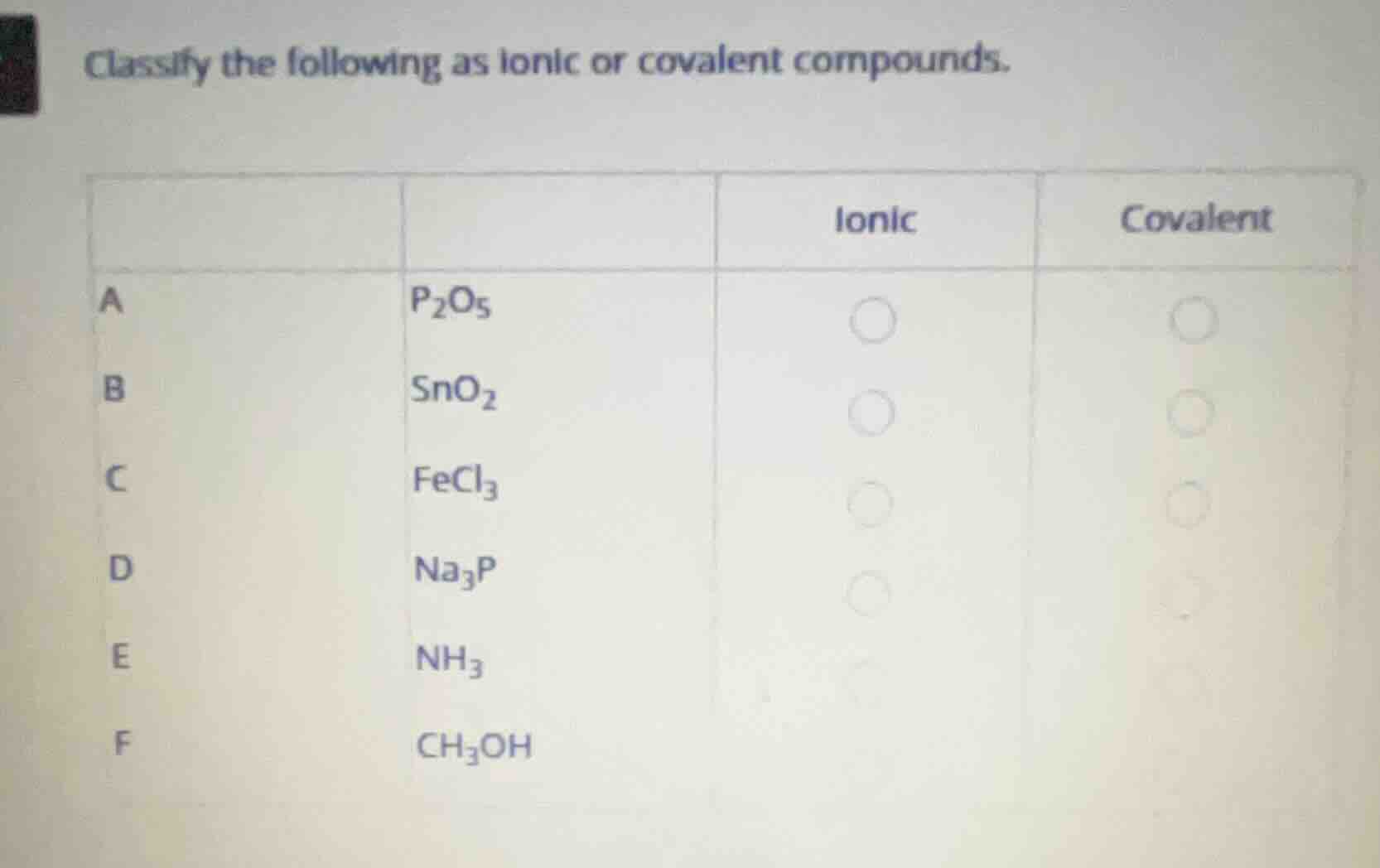
classify the following as ionic or covalent compounds.
a \\(\ce{p2o5}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
b \\(\ce{sno2}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
c \\(\ce{fecl3}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
d \\(\ce{na3p}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
e \\(\ce{nh3}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
f \\(\ce{ch3oh}\\)
ionic \\(\circ\\) covalent \\(\circ\\)
To classify compounds as ionic or covalent, we use the following rules:
- Ionic compounds: Form between a metal (or polyatomic cation) and a non - metal (or polyatomic anion), involving electron transfer.
- Covalent compounds: Form between non - metals, involving electron sharing.
Compound A: $\boldsymbol{P_2O_5}$
Phosphorus ($P$) and oxygen ($O$) are both non - metals. So, they share electrons to form a covalent compound.
Classification: Covalent
Compound B: $\boldsymbol{SnO_2}$
Tin ($Sn$) is a metal, and oxygen ($O$) is a non - metal. The metal (Sn) and non - metal (O) transfer electrons, so it is an ionic compound.
Classification: Ionic
Compound C: $\boldsymbol{FeCl_3}$
Iron ($Fe$) is a metal, and chlorine ($Cl$) is a non - metal. There is electron transfer between the metal (Fe) and non - metal (Cl), so it is an ionic compound.
Classification: Ionic
Compound D: $\boldsymbol{Na_3P}$
Sodium ($Na$) is a metal, and phosphorus ($P$) is a non - metal. Electron transfer occurs between the metal (Na) and non - metal (P), so it is an ionic compound.
Classification: Ionic
Compound E: $\boldsymbol{NH_3}$
Nitrogen ($N$) and hydrogen ($H$) are non - metals. They share electrons to form a covalent compound.
Classification: Covalent
Compound F: $\boldsymbol{CH_3OH}$ (Methanol)
Carbon ($C$), hydrogen ($H$), and oxygen ($O$) are non - metals. The atoms share electrons, so it is a covalent compound.
Classification: Covalent
Final Classifications:
| Compound | Ionic | Covalent |
|---|---|---|
| B ($SnO_2$) | $\boldsymbol{\bigcirc}$ | ∘ |
| C ($FeCl_3$) | $\boldsymbol{\bigcirc}$ | ∘ |
| D ($Na_3P$) | $\boldsymbol{\bigcirc}$ | ∘ |
| E ($NH_3$) | ∘ | $\boldsymbol{\bigcirc}$ |
| F ($CH_3OH$) | ∘ | $\boldsymbol{\bigcirc}$ |
(Note: In the table, $\bigcirc$ represents the correct classification for each compound.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To classify compounds as ionic or covalent, we use the following rules:
- Ionic compounds: Form between a metal (or polyatomic cation) and a non - metal (or polyatomic anion), involving electron transfer.
- Covalent compounds: Form between non - metals, involving electron sharing.
Compound A: $\boldsymbol{P_2O_5}$
Phosphorus ($P$) and oxygen ($O$) are both non - metals. So, they share electrons to form a covalent compound.
Classification: Covalent
Compound B: $\boldsymbol{SnO_2}$
Tin ($Sn$) is a metal, and oxygen ($O$) is a non - metal. The metal (Sn) and non - metal (O) transfer electrons, so it is an ionic compound.
Classification: Ionic
Compound C: $\boldsymbol{FeCl_3}$
Iron ($Fe$) is a metal, and chlorine ($Cl$) is a non - metal. There is electron transfer between the metal (Fe) and non - metal (Cl), so it is an ionic compound.
Classification: Ionic
Compound D: $\boldsymbol{Na_3P}$
Sodium ($Na$) is a metal, and phosphorus ($P$) is a non - metal. Electron transfer occurs between the metal (Na) and non - metal (P), so it is an ionic compound.
Classification: Ionic
Compound E: $\boldsymbol{NH_3}$
Nitrogen ($N$) and hydrogen ($H$) are non - metals. They share electrons to form a covalent compound.
Classification: Covalent
Compound F: $\boldsymbol{CH_3OH}$ (Methanol)
Carbon ($C$), hydrogen ($H$), and oxygen ($O$) are non - metals. The atoms share electrons, so it is a covalent compound.
Classification: Covalent
Final Classifications:
| Compound | Ionic | Covalent |
|---|---|---|
| B ($SnO_2$) | $\boldsymbol{\bigcirc}$ | ∘ |
| C ($FeCl_3$) | $\boldsymbol{\bigcirc}$ | ∘ |
| D ($Na_3P$) | $\boldsymbol{\bigcirc}$ | ∘ |
| E ($NH_3$) | ∘ | $\boldsymbol{\bigcirc}$ |
| F ($CH_3OH$) | ∘ | $\boldsymbol{\bigcirc}$ |
(Note: In the table, $\bigcirc$ represents the correct classification for each compound.)