QUESTION IMAGE
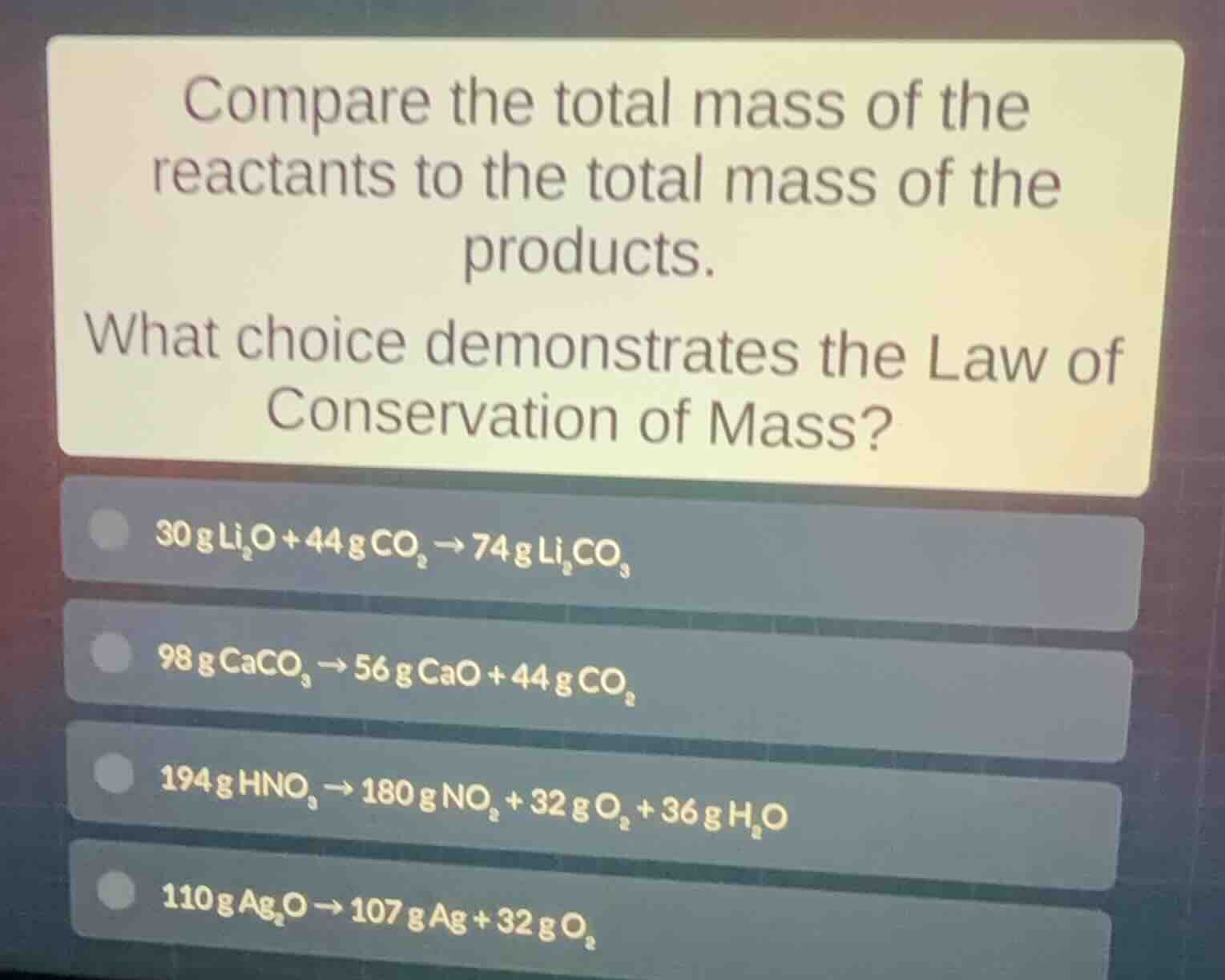
Question
compare the total mass of the reactants to the total mass of the products. what choice demonstrates the law of conservation of mass? 30g li₂o + 44g co₂ → 74g li₂co₃ 98g caco₃ → 56g cao + 44g co₂ 194g hno₃ → 180g no₂ + 32g o₂ + 36g h₂o 110g ag₂o → 107g ag + 32g o₂
Step1: Calculate reactant mass (Option1)
$30\ \text{g} + 44\ \text{g} = 74\ \text{g}$
Step2: Compare to product mass (Option1)
$74\ \text{g}
eq 74\ \text{g}$ (Incorrect, sum mismatch)
Step3: Calculate reactant mass (Option2)
$98\ \text{g}$
Step4: Compare to product mass (Option2)
$56\ \text{g} + 44\ \text{g} = 100\ \text{g}$, $98\ \text{g}
eq 100\ \text{g}$ (Incorrect)
Step5: Calculate reactant mass (Option3)
$194\ \text{g}$
Step6: Compare to product mass (Option3)
$180\ \text{g} + 32\ \text{g} + 36\ \text{g} = 248\ \text{g}$, $194\ \text{g}
eq 248\ \text{g}$ (Incorrect)
Step7: Calculate reactant mass (Option4)
$110\ \text{g}$
Step8: Compare to product mass (Option4)
$107\ \text{g} + 32\ \text{g} = 139\ \text{g}$, $110\ \text{g}
eq 139\ \text{g}$ (Incorrect)
Correction: Recheck Option1 calculation error
Step9: Recalculate Option1 reactant total
$30 + 44 = 74\ \text{g}$, matches product $74\ \text{g}$ (Correct)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $30\ \text{g}\ \text{Li}_2\text{O} + 44\ \text{g}\ \text{CO}_2
ightarrow 74\ \text{g}\ \text{Li}_2\text{CO}_3$