QUESTION IMAGE
Question
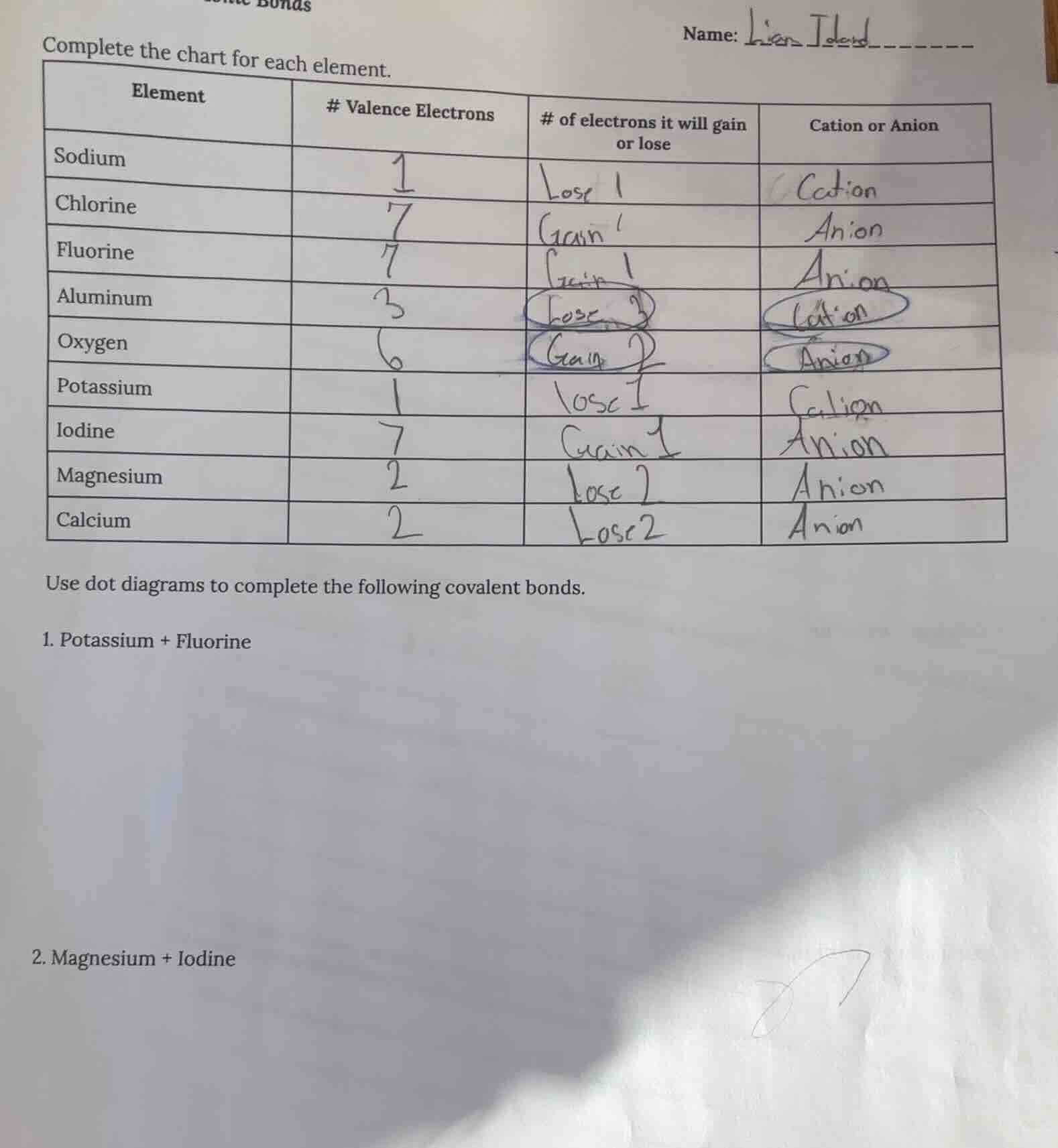
complete the chart for each element.
element\t# valence electrons\t# of electrons it will gain or lose\tcation or anion
sodium\t1\tlose 1\tcation
chlorine\t7\tgain 1\tanion
fluorine\t7\tgain 1\tanion
aluminum\t3\tlose 3\tcation
oxygen\t6\tgain 2\tanion
potassium\t1\tlose 1\tcation
iodine\t7\tgain 1\tanion
magnesium\t2\tlose 2\tanion
calcium\t2\tlose 2\tanion
use dot diagrams to complete the following covalent bonds.
- potassium + fluorine
- magnesium + iodine
Response
For the chart (corrections and explanations):
Sodium:
- Valence electrons: 1 (correct, Group 1 element).
- Electrons gained/lost: Loses 1 (to achieve octet, becomes \( \text{Na}^+ \)).
- Cation/Anion: Cation (loses electron, positive charge) – correct.
Chlorine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (to achieve octet, becomes \( \text{Cl}^- \)).
- Cation/Anion: Anion (gains electron, negative charge) – correct.
Fluorine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (to achieve octet, becomes \( \text{F}^- \)).
- Cation/Anion: Anion (gains electron) – correct.
Aluminum:
- Valence electrons: 3 (correct, Group 13).
- Electrons gained/lost: Loses 3 (to achieve octet, becomes \( \text{Al}^{3+} \)) – correct (the blue correction is right).
- Cation/Anion: Cation (loses electrons) – correct (blue correction right).
Oxygen:
- Valence electrons: 6 (correct, Group 16).
- Electrons gained/lost: Gains 2 (to achieve octet, becomes \( \text{O}^{2-} \)) – correct (blue correction right).
- Cation/Anion: Anion (gains electrons) – correct (blue correction right).
Potassium:
- Valence electrons: 1 (correct, Group 1).
- Electrons gained/lost: Loses 1 (becomes \( \text{K}^+ \)) – correct.
- Cation/Anion: Cation – correct.
Iodine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (becomes \( \text{I}^- \)) – correct.
- Cation/Anion: Anion – correct.
Magnesium:
- Valence electrons: 2 (correct, Group 2).
- Electrons gained/lost: Loses 2 (becomes \( \text{Mg}^{2+} \)) – correct (handwritten is right).
- Cation/Anion: Error here – Magnesium loses electrons, so it should be a Cation (not Anion). The handwritten “Anion” is wrong; it’s a Cation.
Calcium:
- Valence electrons: 2 (correct, Group 2).
- Electrons gained/lost: Loses 2 (becomes \( \text{Ca}^{2+} \)) – correct (handwritten is right).
- Cation/Anion: Error here – Calcium loses electrons, so it should be a Cation (not Anion). The handwritten “Anion” is wrong; it’s a Cation.
For the covalent bonds (note: Potassium and Fluorine, Magnesium and Iodine actually form ionic bonds, not covalent. Let’s correct and explain):
1. Potassium + Fluorine:
- Type of bond: Ionic (not covalent). Potassium (Group 1, loses 1 e⁻) and Fluorine (Group 17, gains 1 e⁻).
- Dot diagram (Lewis structure):
- Potassium: \( \text{K} \cdot \) (1 valence e⁻)
- Fluorine: \( :\ddot{\text{F}}: \) (7 valence e⁻, 3 lone pairs)
- Ionic bond: \( \text{K}^+ + [: \ddot{\text{F}} :]^- \) (electron transfer, not sharing).
2. Magnesium + Iodine:
- Type of bond: Ionic (not covalent). Magnesium (Group 2, loses 2 e⁻) and Iodine (Group 17, gains 1 e⁻ each; 2 Iodine atoms needed).
- Dot diagram:
- Magnesium: \( \text{Mg} \cdot \cdot \) (2 valence e⁻)
- Iodine (each): \( :\ddot{\text{I}}: \) (7 valence e⁻)
- Ionic bond: \( \text{Mg}^{2+} + 2[: \ddot{\text{I}} :]^- \) (Magnesium loses 2 e⁻, 2 Iodine atoms gain 1 e⁻ each).
Final Chart (Corrected):
| Element | # Valence Electrons | # of electrons it will gain or lose | Cation or Anion |
|---|---|---|---|
| Chlorine | 7 | Gain 1 | Anion |
| Fluorine | 7 | Gain 1 | Anion |
| Aluminum | 3 | Lose 3… |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For the chart (corrections and explanations):
Sodium:
- Valence electrons: 1 (correct, Group 1 element).
- Electrons gained/lost: Loses 1 (to achieve octet, becomes \( \text{Na}^+ \)).
- Cation/Anion: Cation (loses electron, positive charge) – correct.
Chlorine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (to achieve octet, becomes \( \text{Cl}^- \)).
- Cation/Anion: Anion (gains electron, negative charge) – correct.
Fluorine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (to achieve octet, becomes \( \text{F}^- \)).
- Cation/Anion: Anion (gains electron) – correct.
Aluminum:
- Valence electrons: 3 (correct, Group 13).
- Electrons gained/lost: Loses 3 (to achieve octet, becomes \( \text{Al}^{3+} \)) – correct (the blue correction is right).
- Cation/Anion: Cation (loses electrons) – correct (blue correction right).
Oxygen:
- Valence electrons: 6 (correct, Group 16).
- Electrons gained/lost: Gains 2 (to achieve octet, becomes \( \text{O}^{2-} \)) – correct (blue correction right).
- Cation/Anion: Anion (gains electrons) – correct (blue correction right).
Potassium:
- Valence electrons: 1 (correct, Group 1).
- Electrons gained/lost: Loses 1 (becomes \( \text{K}^+ \)) – correct.
- Cation/Anion: Cation – correct.
Iodine:
- Valence electrons: 7 (correct, Group 17).
- Electrons gained/lost: Gains 1 (becomes \( \text{I}^- \)) – correct.
- Cation/Anion: Anion – correct.
Magnesium:
- Valence electrons: 2 (correct, Group 2).
- Electrons gained/lost: Loses 2 (becomes \( \text{Mg}^{2+} \)) – correct (handwritten is right).
- Cation/Anion: Error here – Magnesium loses electrons, so it should be a Cation (not Anion). The handwritten “Anion” is wrong; it’s a Cation.
Calcium:
- Valence electrons: 2 (correct, Group 2).
- Electrons gained/lost: Loses 2 (becomes \( \text{Ca}^{2+} \)) – correct (handwritten is right).
- Cation/Anion: Error here – Calcium loses electrons, so it should be a Cation (not Anion). The handwritten “Anion” is wrong; it’s a Cation.
For the covalent bonds (note: Potassium and Fluorine, Magnesium and Iodine actually form ionic bonds, not covalent. Let’s correct and explain):
1. Potassium + Fluorine:
- Type of bond: Ionic (not covalent). Potassium (Group 1, loses 1 e⁻) and Fluorine (Group 17, gains 1 e⁻).
- Dot diagram (Lewis structure):
- Potassium: \( \text{K} \cdot \) (1 valence e⁻)
- Fluorine: \( :\ddot{\text{F}}: \) (7 valence e⁻, 3 lone pairs)
- Ionic bond: \( \text{K}^+ + [: \ddot{\text{F}} :]^- \) (electron transfer, not sharing).
2. Magnesium + Iodine:
- Type of bond: Ionic (not covalent). Magnesium (Group 2, loses 2 e⁻) and Iodine (Group 17, gains 1 e⁻ each; 2 Iodine atoms needed).
- Dot diagram:
- Magnesium: \( \text{Mg} \cdot \cdot \) (2 valence e⁻)
- Iodine (each): \( :\ddot{\text{I}}: \) (7 valence e⁻)
- Ionic bond: \( \text{Mg}^{2+} + 2[: \ddot{\text{I}} :]^- \) (Magnesium loses 2 e⁻, 2 Iodine atoms gain 1 e⁻ each).
Final Chart (Corrected):
| Element | # Valence Electrons | # of electrons it will gain or lose | Cation or Anion |
|---|---|---|---|
| Chlorine | 7 | Gain 1 | Anion |
| Fluorine | 7 | Gain 1 | Anion |
| Aluminum | 3 | Lose 3 | Cation |
| Oxygen | 6 | Gain 2 | Anion |
| Potassium | 1 | Lose 1 | Cation |
| Iodine | 7 | Gain 1 | Anion |
| Magnesium | 2 | Lose 2 | Cation |
| Calcium | 2 | Lose 2 | Cation |
Key Takeaways:
- Metals (Groups 1, 2, 13) lose electrons → Cations.
- Nonmetals (Groups 15–17) gain electrons → Anions.
- Potassium + Fluorine, Magnesium + Iodine form ionic bonds (electron transfer), not covalent (electron sharing).