QUESTION IMAGE
Question
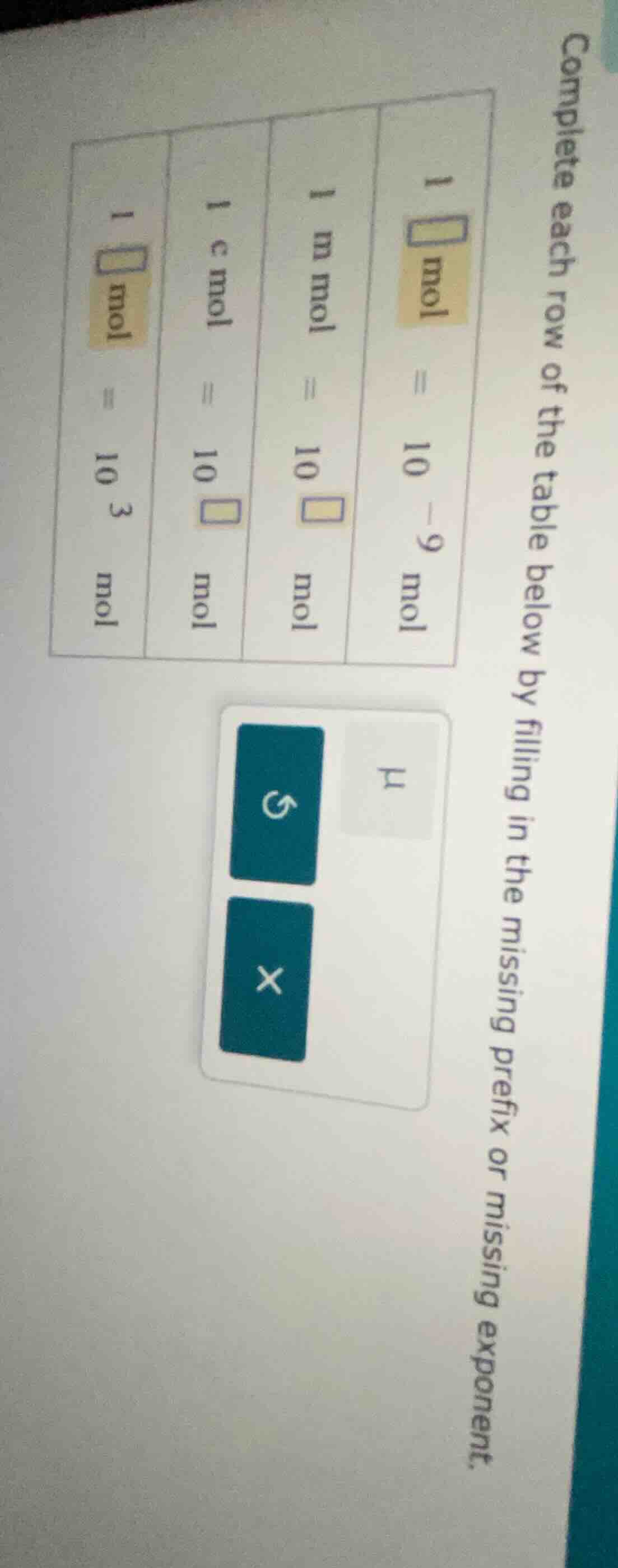
complete each row of the table below by filling in the missing prefix or missing exponent.
1 \boxed{} mol = 10^{-9} mol
1 mmol = 10^{\boxed{}} mol
1 cmol = 10^{\boxed{}} mol
1 \boxed{} mol = 10^{3} mol
Step1: Recall metric prefixes
Metric prefixes and their corresponding powers of 10:
- nano (n) = \(10^{-9}\)
- micro (μ) = \(10^{-6}\)
- milli (m) = \(10^{-3}\)
- centi (c) = \(10^{-2}\)
- kilo (k) = \(10^{3}\), but here we have 1 [prefix] mol = \(10^3\) mol, wait no, wait the first row: 1 [prefix] mol = \(10^{-9}\) mol. The prefix for \(10^{-9}\) is nano (n), but wait the options? Wait the given prefixes: let's check each row.
First row: 1 [prefix] mol = \(10^{-9}\) mol. The prefix for \(10^{-9}\) is nano (n), but wait the button has μ (micro, \(10^{-6}\)), m (milli, \(10^{-3}\)), c (centi, \(10^{-2}\)), k (kilo, \(10^3\))? Wait no, the first row: 1 [prefix] mol = \(10^{-9}\) mol. The prefix for \(10^{-9}\) is nano, but maybe the problem has a typo? Wait no, maybe I misread. Wait the first row: 1 [blank] mol = \(10^{-9}\) mol. The prefix for \(10^{-9}\) is nano (n), but the options given? Wait the buttons: μ (micro, \(10^{-6}\)), m (milli, \(10^{-3}\)), c (centi, \(10^{-2}\)), and the other is a reset or cross. Wait maybe the first row: 1 nmol = \(10^{-9}\) mol, but the options? Wait no, maybe the first row's prefix is nano, but the given options? Wait the second row: 1 mmol = \(10^{?}\) mol. Milli is \(10^{-3}\), so 1 mmol = \(10^{-3}\) mol. Third row: 1 cmol = \(10^{?}\) mol. Centi is \(10^{-2}\), so 1 cmol = \(10^{-2}\) mol. Fourth row: 1 [prefix] mol = \(10^{3}\) mol. Kilo is \(10^3\), so 1 kmol = \(10^3\) mol, but the prefix here? Wait the fourth row: 1 [blank] mol = \(10^3\) mol, so the prefix is kilo (k), but the given buttons? Wait the problem's table:
Row 1: 1 [prefix] mol = \(10^{-9}\) mol. The prefix for \(10^{-9}\) is nano (n), but the options? Wait the button has μ (micro, \(10^{-6}\)), m (milli, \(10^{-3}\)), c (centi, \(10^{-2}\)). Wait maybe the first row is 1 nmol = \(10^{-9}\) mol, but the options? Wait no, maybe I made a mistake. Wait let's do each row:
Row 1: 1 [prefix] mol = \(10^{-9}\) mol. The prefix is nano (n), but the given options? Wait the second row: 1 mmol = \(10^{?}\) mol. Milli is \(10^{-3}\), so exponent is -3.
Row 3: 1 cmol = \(10^{?}\) mol. Centi is \(10^{-2}\), so exponent is -2.
Row 4: 1 [prefix] mol = \(10^{3}\) mol. Kilo is \(10^3\), so prefix is kilo (k), but the options? Wait the fourth row's prefix is kilo, so 1 kmol = \(10^3\) mol.
Wait let's re-express:
- 1 [nano] mol = \(10^{-9}\) mol (prefix: n, exponent: -9)
- 1 mmol = \(10^{-3}\) mol (exponent: -3)
- 1 cmol = \(10^{-2}\) mol (exponent: -2)
- 1 kmol = \(10^{3}\) mol (prefix: k, exponent: 3)
But the problem's first row: 1 [blank] mol = \(10^{-9}\) mol. The prefix is nano (n), so the blank is n, but the options given? Wait the buttons: μ (micro, \(10^{-6}\)), m (milli, \(10^{-3}\)), c (centi, \(10^{-2}\)), and the other is a cross or reset. Wait maybe the first row's prefix is nano, but the problem has a typo, or I misread. Wait the first row's exponent is -9, so the prefix is nano (n), but the options? Wait the second row: 1 mmol = \(10^{?}\) mol. Milli is \(10^{-3}\), so exponent is -3.
Third row: 1 cmol = \(10^{?}\) mol. Centi is \(10^{-2}\), so exponent is -2.
Fourth row: 1 [prefix] mol = \(10^3\) mol. Kilo is \(10^3\), so prefix is k, exponent 3.
So let's fill each row:
- 1 nmol = \(10^{-9}\) mol (prefix: n, exponent: -9)
- 1 mmol = \(10^{-3}\) mol (exponent: -3)
- 1 cmol = \(10^{-2}\) mol (exponent: -2)
- 1 kmol = \(10^{3}\) mol (prefix: k, exponent: 3)
But the problem's first row: the blank is the prefix, so 1 nmol = \(10^{-9}\) mol. The second row: 1 mmol = \(10^{-3}\) mol, so exponent is -3. Third row: 1 c…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First row: prefix = n (nano), so 1 nmol = \(10^{-9}\) mol
Second row: exponent = -3, so 1 mmol = \(10^{-3}\) mol
Third row: exponent = -2, so 1 cmol = \(10^{-2}\) mol
Fourth row: prefix = k (kilo), so 1 kmol = \(10^{3}\) mol
(Assuming the first row's prefix is nano, second row's exponent -3, third row's exponent -2, fourth row's prefix kilo)