QUESTION IMAGE
Question
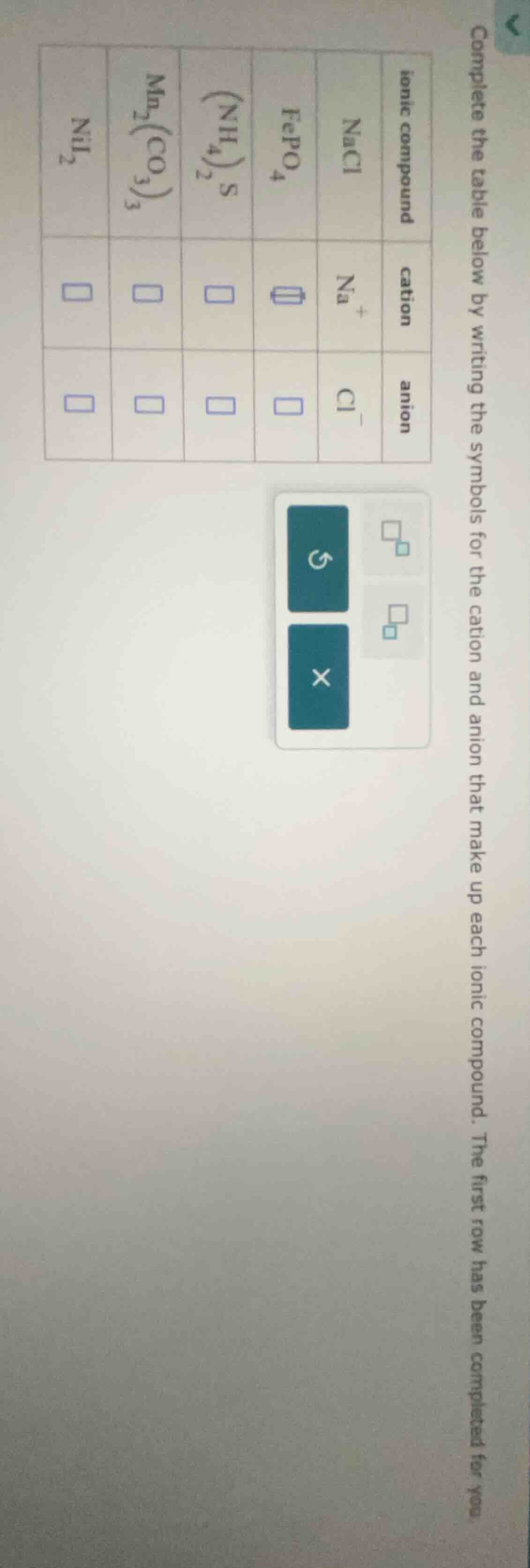
complete the table below by writing the symbols for the cation and anion that make up each ionic compound. the first row has been completed for you.
| ionic compound | nacl | fepo₄ | (nh₄)₂s | mn₂(co₃)₃ | nii₂ |
|---|---|---|---|---|---|
| anion | cl⁻ |
Step1: Analyze \( \text{FePO}_4 \)
In \( \text{FePO}_4 \), the phosphate ion is \( \text{PO}_4^{3-} \). To balance the charge, iron must have a \( +3 \) charge (since \( (+3) + (-3) = 0 \)). So the cation is \( \text{Fe}^{3+} \) and the anion is \( \text{PO}_4^{3-} \).
Step2: Analyze \( (\text{NH}_4)_2\text{S} \)
Ammonium ion is \( \text{NH}_4^+ \), and sulfide ion is \( \text{S}^{2-} \). The formula has 2 ammonium ions to balance the \( -2 \) charge of sulfide. So cation is \( \text{NH}_4^+ \), anion is \( \text{S}^{2-} \).
Step3: Analyze \( \text{Mn}_2(\text{CO}_3)_3 \)
Carbonate ion is \( \text{CO}_3^{2-} \). There are 3 carbonate ions (total charge \( 3\times(-2) = -6 \)), so 2 Mn ions must have total charge \( +6 \), meaning each Mn has \( +3 \) ( \( \text{Mn}^{3+} \) ). Anion is \( \text{CO}_3^{2-} \).
Step4: Analyze \( \text{NiI}_2 \)
Iodide ion is \( \text{I}^- \). There are 2 iodide ions (total charge \( -2 \)), so Ni has \( +2 \) charge ( \( \text{Ni}^{2+} \) ). Anion is \( \text{I}^- \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Ionic Compound | Cation | Anion |
|---|---|---|
| \( \text{FePO}_4 \) | \( \text{Fe}^{3+} \) | \( \text{PO}_4^{3-} \) |
| \( (\text{NH}_4)_2\text{S} \) | \( \text{NH}_4^+ \) | \( \text{S}^{2-} \) |
| \( \text{Mn}_2(\text{CO}_3)_3 \) | \( \text{Mn}^{3+} \) | \( \text{CO}_3^{2-} \) |
| \( \text{NiI}_2 \) | \( \text{Ni}^{2+} \) | \( \text{I}^- \) |