QUESTION IMAGE
Question
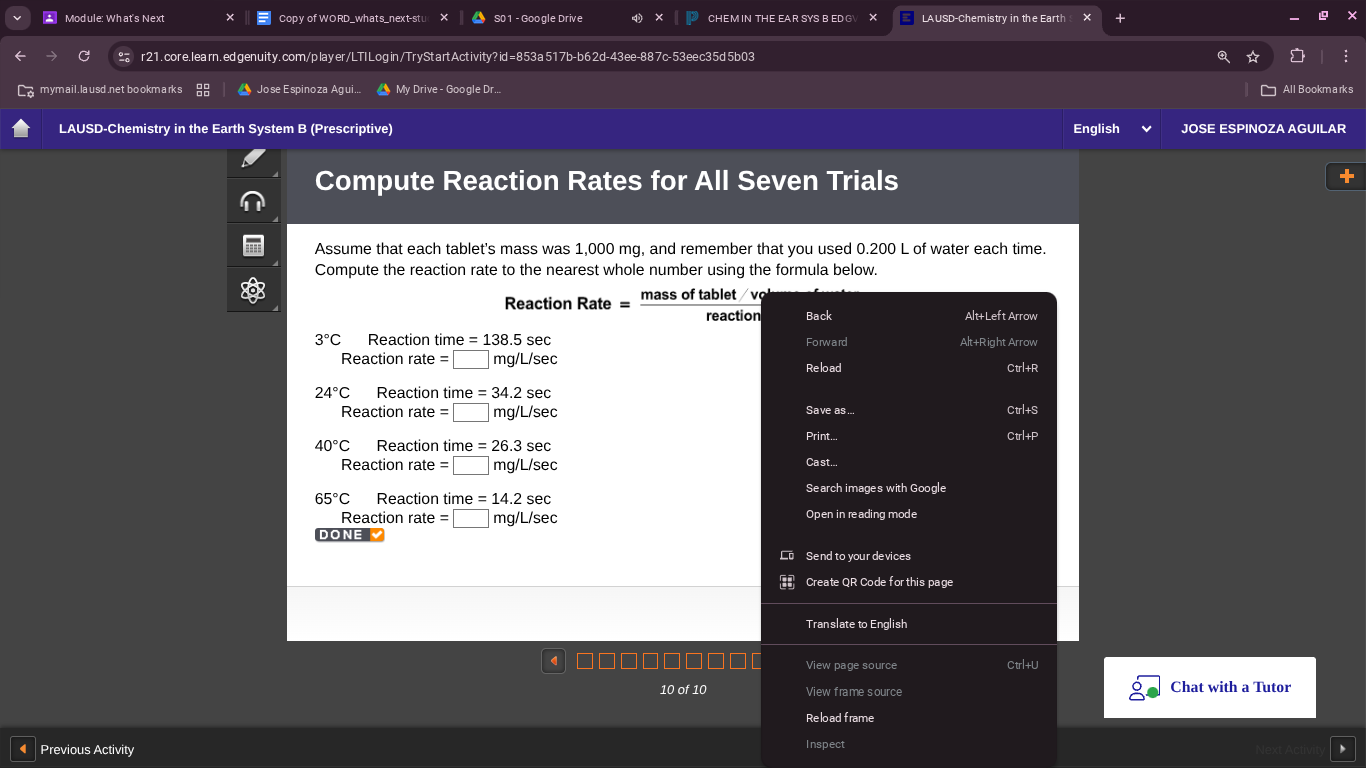
compute reaction rates for all seven trials
assume that each tablets mass was 1,000 mg, and remember that you used 0.200 l of water each time. compute the reaction rate to the nearest whole number using the formula below.
reaction rate =
3°c reaction time = 138.5 sec
reaction rate = mg/l/sec
24°c reaction time = 34.2 sec
reaction rate = mg/l/sec
40°c reaction time = 26.3 sec
reaction rate = mg/l/sec
65°c reaction time = 14.2 sec
reaction rate = mg/l/sec
Step1: Recall the reaction rate formula
The formula for reaction rate is \( \text{Reaction Rate} = \frac{\text{mass of tablet}}{\text{volume of water} \times \text{reaction time}} \). Given mass of tablet \( = 1000 \, \text{mg} \), volume of water \( = 0.200 \, \text{L} \).
Step2: Calculate for \( 3^\circ \text{C} \)
Reaction time \( = 138.5 \, \text{sec} \).
Substitute into formula: \( \text{Reaction Rate} = \frac{1000}{0.200 \times 138.5} \)
First, calculate denominator: \( 0.200 \times 138.5 = 27.7 \)
Then, \( \frac{1000}{27.7} \approx 36 \) (rounded to nearest whole number)
Step3: Calculate for \( 24^\circ \text{C} \)
Reaction time \( = 34.2 \, \text{sec} \).
Denominator: \( 0.200 \times 34.2 = 6.84 \)
Reaction Rate: \( \frac{1000}{6.84} \approx 146 \)
Step4: Calculate for \( 40^\circ \text{C} \)
Reaction time \( = 26.3 \, \text{sec} \).
Denominator: \( 0.200 \times 26.3 = 5.26 \)
Reaction Rate: \( \frac{1000}{5.26} \approx 190 \)
Step5: Calculate for \( 65^\circ \text{C} \)
Reaction time \( = 14.2 \, \text{sec} \).
Denominator: \( 0.200 \times 14.2 = 2.84 \)
Reaction Rate: \( \frac{1000}{2.84} \approx 352 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For \( 3^\circ \text{C} \): 36
For \( 24^\circ \text{C} \): 146
For \( 40^\circ \text{C} \): 190
For \( 65^\circ \text{C} \): 352