QUESTION IMAGE
Question
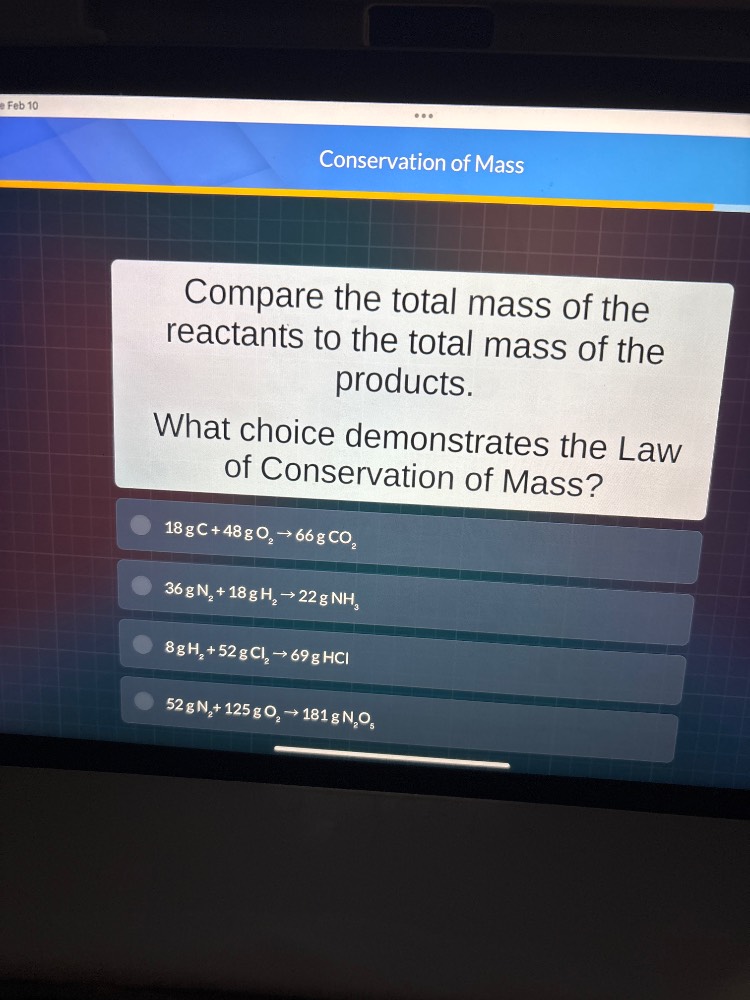
conservation of mass
compare the total mass of the reactants to the total mass of the products.
what choice demonstrates the law of conservation of mass?
18 g c + 48 g o₂ → 66 g co₂
36 g n₂ + 18 g h₂ → 22 g nh₃
8 g h₂ + 52 g cl₂ → 69 g hcl
52 g n₂ + 125 g o₂ → 181 g n₂o₅
Step1: Calculate reactant mass (1st option)
$18\ \text{g} + 48\ \text{g} = 66\ \text{g}$
Step2: Compare to product mass (1st option)
Reactant mass ($66\ \text{g}$) = Product mass ($66\ \text{g}$)
Step3: Calculate reactant mass (2nd option)
$36\ \text{g} + 18\ \text{g} = 54\ \text{g}$
Step4: Compare to product mass (2nd option)
$54\ \text{g}
eq 22\ \text{g}$
Step5: Calculate reactant mass (3rd option)
$8\ \text{g} + 52\ \text{g} = 60\ \text{g}$
Step6: Compare to product mass (3rd option)
$60\ \text{g}
eq 69\ \text{g}$
Step7: Calculate reactant mass (4th option)
$52\ \text{g} + 125\ \text{g} = 177\ \text{g}$
Step8: Compare to product mass (4th option)
$177\ \text{g}
eq 181\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $18\ \text{g C} + 48\ \text{g O}_2
ightarrow 66\ \text{g CO}_2$