QUESTION IMAGE
Question
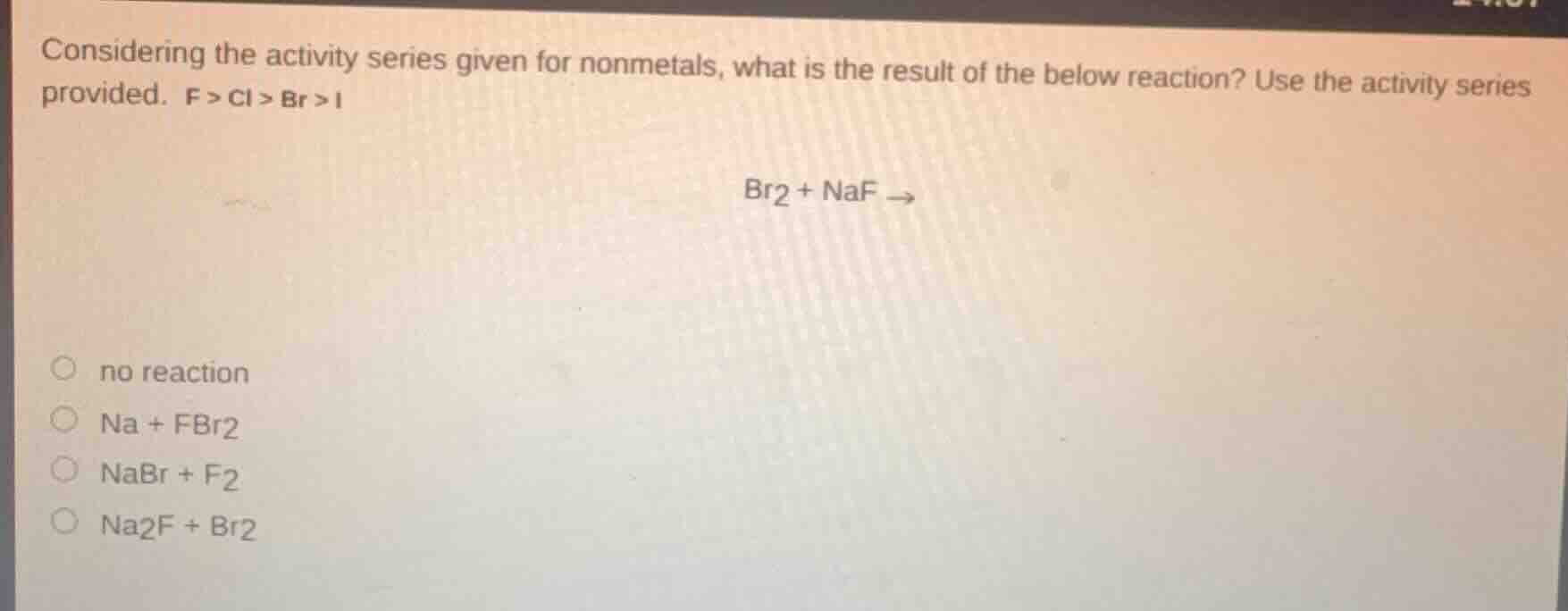
considering the activity series given for nonmetals, what is the result of the below reaction? use the activity series provided. f > cl > br > i
br₂ + naf →
○ no reaction
○ na + fbr₂
○ nabr + f₂
○ na₂f + br₂
Step1: Recall activity series rule
For nonmetal displacement reactions, a more active nonmetal can displace a less active nonmetal from its compound. The activity series given is \( F > Cl > Br > I \), meaning F is more active than Br.
Step2: Analyze the reaction
The reaction is \( \text{Br}_2 + \text{NaF}
ightarrow \). Here, Br (in \( \text{Br}_2 \)) is less active than F (in \( \text{NaF} \)) according to the activity series (\( F > Br \)). So, Br cannot displace F from \( \text{NaF} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
no reaction