QUESTION IMAGE
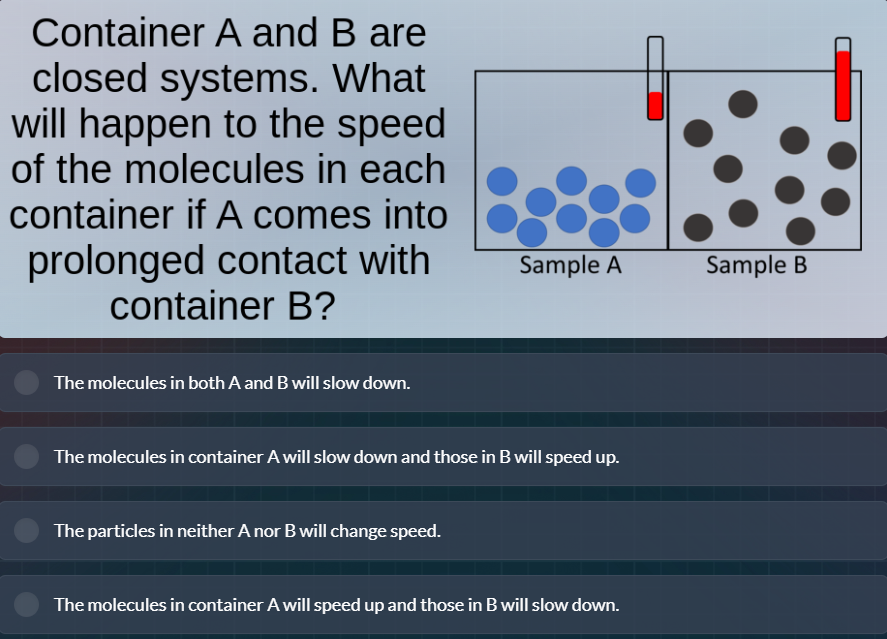
Question
container a and b are closed systems. what will happen to the speed of the molecules in each container if a comes into prolonged contact with container b?
the molecules in both a and b will slow down.
the molecules in container a will slow down and those in b will speed up.
the particles in neither a nor b will change speed.
the molecules in container a will speed up and those in b will slow down.
In a closed system, when two containers (A and B) with different molecular speeds (implied by the different thermometer readings, with B having higher temperature initially as the thermometer is more red) are in prolonged contact, heat transfer occurs until thermal equilibrium. Molecules in the hotter system (B) have higher kinetic energy (faster speed), and those in the colder system (A) have lower kinetic energy (slower speed). Heat flows from B to A. So, molecules in B (losing heat) slow down, and molecules in A (gaining heat) speed up.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The molecules in container A will speed up and those in B will slow down.