QUESTION IMAGE
Question
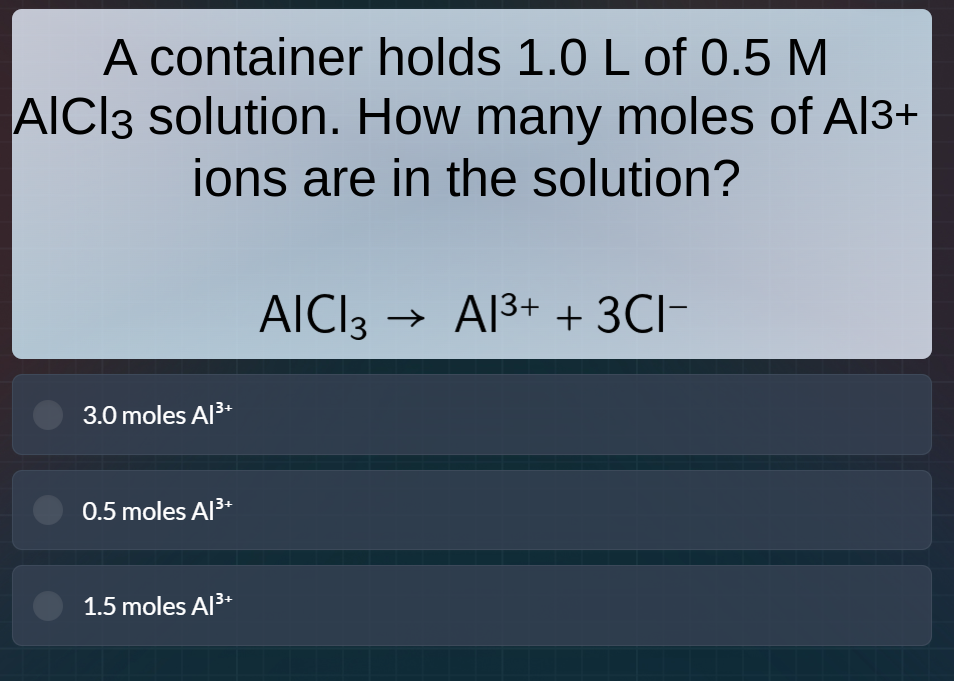
a container holds 1.0 l of 0.5 m alcl₃ solution. how many moles of al³⁺ ions are in the solution? alcl₃ → al³⁺ + 3cl⁻ 3.0 moles al³⁺ 0.5 moles al³⁺ 1.5 moles al³⁺
Step1: Calculate moles of \( AlCl_3 \)
Molarity formula: \( M = \frac{n}{V} \), so \( n = M \times V \).
Given \( M = 0.5 \, M \), \( V = 1.0 \, L \),
\( n_{AlCl_3} = 0.5 \, \frac{mol}{L} \times 1.0 \, L = 0.5 \, mol \).
Step2: Relate \( AlCl_3 \) to \( Al^{3+} \)
From dissociation: \( 1 \, mol \, AlCl_3
ightarrow 1 \, mol \, Al^{3+} \).
Thus, \( n_{Al^{3+}} = n_{AlCl_3} = 0.5 \, mol \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.5 moles \( \text{Al}^{3+} \) (corresponding to the option: 0.5 moles \( \text{Al}^{3+} \))