QUESTION IMAGE
Question
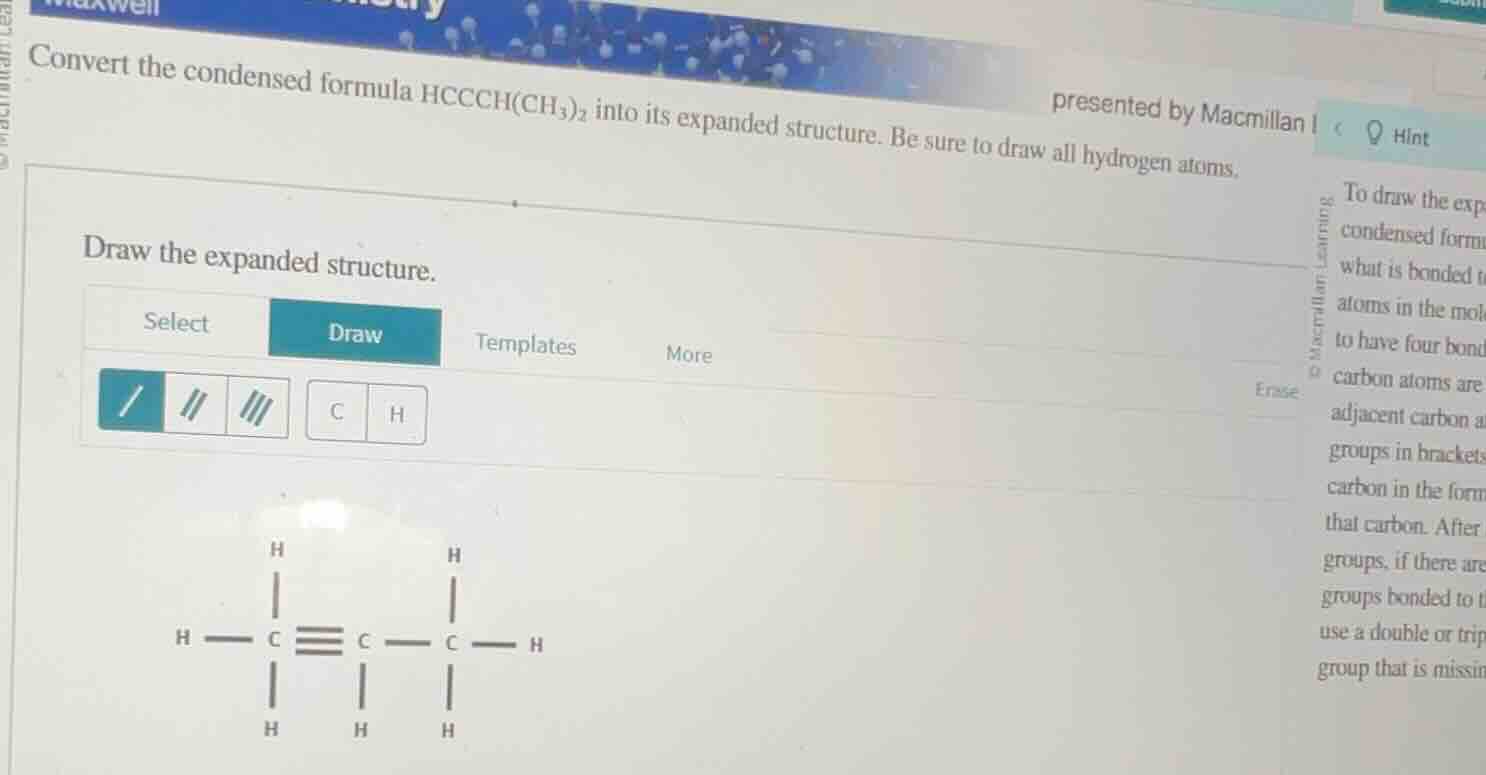
convert the condensed formula hccch(ch₃)₂ into its expanded structure. be sure to draw all hydrogen atoms. draw the expanded structure. select draw templates more / || ||| c h h h c ≡ c c h h h h presented by macmillan hint to draw the exp condensed form what is bonded t atoms in the mol to have four bond carbon atoms are adjacent carbon a groups in brackets carbon in the form that carbon. after groups, if there are groups bonded to use a double or trip group that is missin
Step1: Identify carbon skeleton
The condensed formula $\text{HCCCH(CH}_3\text{)}_2$ has 4 total carbons: a chain of 3 carbons ($\text{H-C-C-C-}$) plus one carbon from each of the two $\text{CH}_3$ groups bonded to the 3rd carbon in the chain.
Step2: Add triple bond
The first two carbons ($\text{HC}\equiv\text{C-}$) have a triple bond to satisfy their valency.
Step3: Add hydrogen atoms
- 1st carbon: Bonds to 1 H and has a triple bond to C2, so total bonds: $1 + 3 = 4$
- 2nd carbon: Triple bond to C1, single bond to C3, total bonds: $3 + 1 = 4$
- 3rd carbon: Single bond to C2, single bonds to two $\text{CH}_3$ groups, total bonds: $1 + 2 = 3$, so it needs 1 more H
- Each $\text{CH}_3$ carbon: Bonds to 3 H atoms and 1 bond to C3, total bonds: $3 + 1 = 4$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
H
|
H-C≡C-C-CH₃
| |
H CH₃Expanded structure with all hydrogens drawn explicitly:
H
|
H - C ≡ C - C - H
| |
H |
CH₃
|
H
|
H - C - H
|
HOr in linear explicit form:
$\text{H}-\text{C} \equiv \text{C}-\text{C}(\text{H})(\text{CH}_3)-\text{CH}_3$ with all hydrogens:
$\text{H}-\text{C}(\text{H}) \equiv \text{C}-\text{C}(\text{H})(\text{C}(\text{H})_3)-\text{C}(\text{H})_3$