QUESTION IMAGE
Question
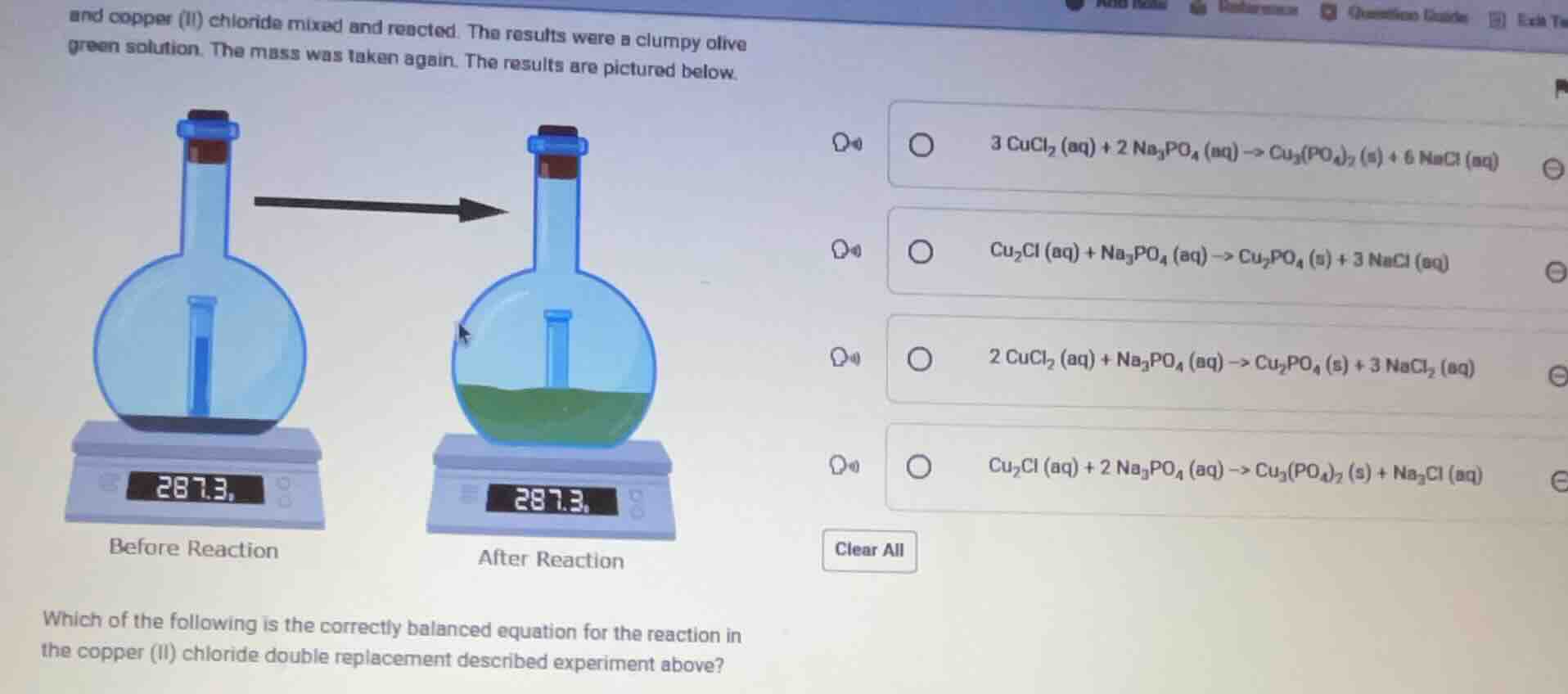
and copper (ii) chloride mixed and reacted. the results were a clumpy olive green solution. the mass was taken again. the results are pictured below.
before reaction
after reaction
which of the following is the correctly balanced equation for the reaction in the copper (ii) chloride double replacement described experiment above?
3 cucl₂ (aq) + 2 na₃po₄ (aq) -> cu₃(po₄)₂ (s) + 6 nacl (aq)
cu₂cl (aq) + na₃po₄ (aq) -> cu₂po₄ (s) + 3 nacl (aq)
2 cucl₂ (aq) + na₃po₄ (aq) -> cu₂po₄ (s) + 3 nacl₂ (aq)
cu₂cl (aq) + 2 na₃po₄ (aq) -> cu₃(po₄)₂ (s) + na₃cl (aq)
Step1: Identify correct reactant formula
Copper (II) chloride is $\text{CuCl}_2$ (since $\text{Cu}^{2+}$ pairs with 2 $\text{Cl}^-$). This eliminates options with $\text{Cu}_2\text{Cl}$.
Step2: Balance copper atoms
Left side: 3 $\text{CuCl}_2$ gives 3 $\text{Cu}^{2+}$. Right side: $\text{Cu}_3(\text{PO}_4)_2$ has 3 $\text{Cu}^{2+}$.
Step3: Balance phosphate atoms
Right side: $\text{Cu}_3(\text{PO}_4)_2$ has 2 $\text{PO}_4^{3-}$. Left side: 2 $\text{Na}_3\text{PO}_4$ gives 2 $\text{PO}_4^{3-}$.
Step4: Balance sodium atoms
Left side: 2 $\text{Na}_3\text{PO}_4$ gives 6 $\text{Na}^+$. Right side: 6 $\text{NaCl}$ gives 6 $\text{Na}^+$.
Step5: Balance chlorine atoms
Left side: 3 $\text{CuCl}_2$ gives 6 $\text{Cl}^-$. Right side: 6 $\text{NaCl}$ gives 6 $\text{Cl}^-$.
All atoms are balanced in this equation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $3\text{CuCl}_2\text{ (aq)} + 2\text{Na}_3\text{PO}_4\text{ (aq)}
ightarrow \text{Cu}_3(\text{PO}_4)_2\text{ (s)} + 6\text{NaCl (aq)}$