QUESTION IMAGE
Question
create complex mathematical models to represent chemical concepts.
ance the following chemical equations
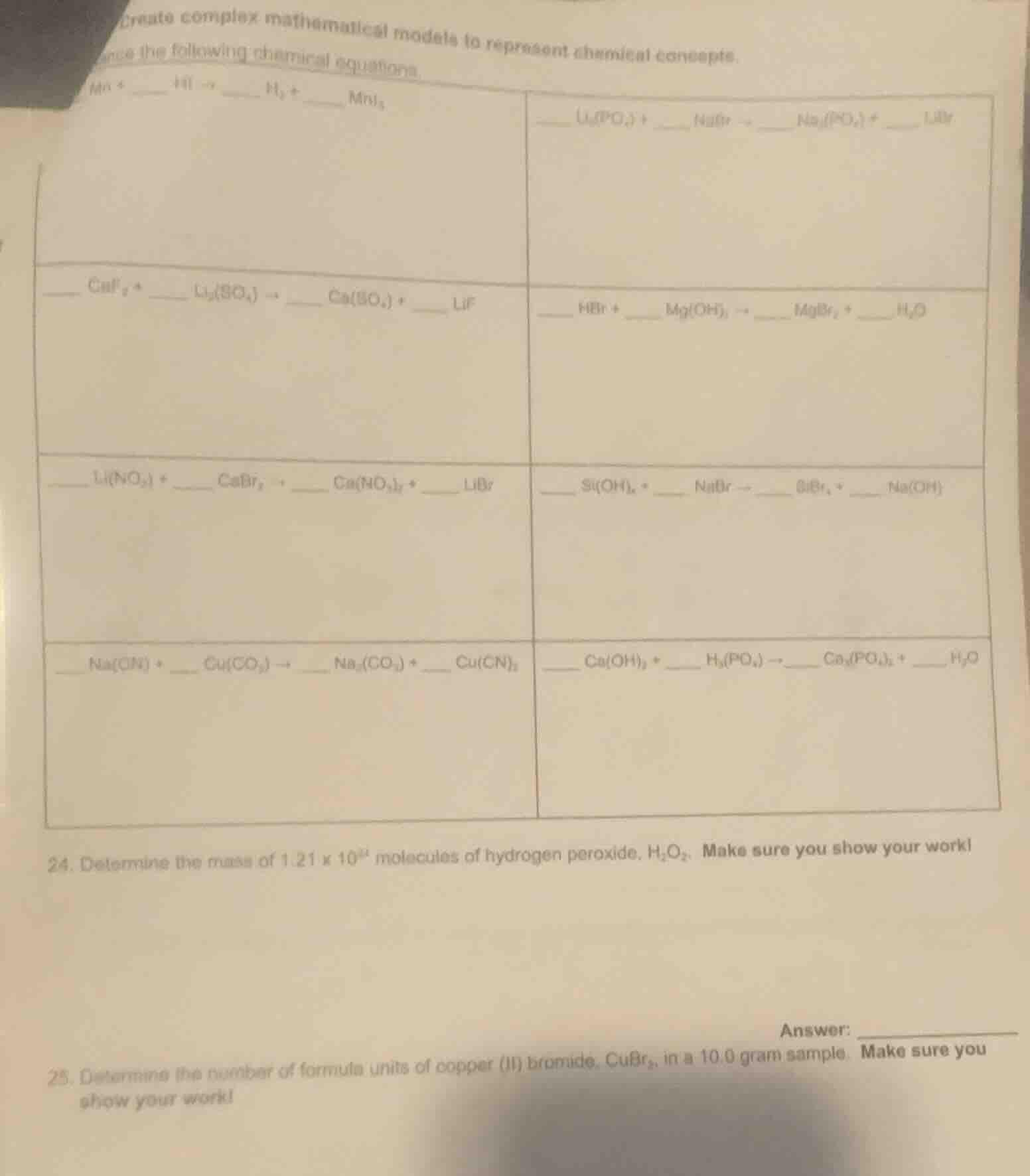
mn + _ hi → _ h₂ + ___ mni₂
_ li₃(po₄) + _ nabr → _ na₃(po₄) + _ libr
_ caf₂ + _ li₂(so₄) → _ ca(so₄) + _ lif
_ hbr + _ mg(oh)₂ → _ mgbr₂ + _ h₂o
_ li(no₃) + _ cabr₂ → _ ca(no₃)₂ + _ libr
_ si(oh)₄ + _ nabr → _ sibr₄ + _ na(oh)
_ na(cn) + _ cu(co₃) → _ na₂(co₃) + _ cu(cn)₂
_ ca(oh)₂ + _ h₃(po₄) → _ ca₃(po₄)₂ + _ h₂o
- determine the mass of 1.21 × 10²⁴ molecules of hydrogen peroxide, h₂o₂. make sure you show your work!
answer: __________
- determine the number of formula units of copper (ii) bromide, cubr₂, in a 10.0 gram sample. make sure you show your work!
Problem 24: Determine the mass of \( 1.21 \times 10^{24} \) molecules of hydrogen peroxide, \( \ce{H_2O_2} \).
Step 1: Find moles of \( \ce{H_2O_2} \)
Use Avogadro's number (\( 6.022 \times 10^{23} \) molecules/mol).
Moles \( = \frac{\text{Number of molecules}}{\text{Avogadro's number}} \)
\( \text{Moles} = \frac{1.21 \times 10^{24}}{6.022 \times 10^{23}} \approx 2.01 \, \text{mol} \)
Step 2: Calculate molar mass of \( \ce{H_2O_2} \)
Molar mass of \( \ce{H} = 1.008 \, \text{g/mol} \), \( \ce{O} = 16.00 \, \text{g/mol} \).
\( \text{Molar mass} = 2(1.008) + 2(16.00) = 2.016 + 32.00 = 34.016 \, \text{g/mol} \approx 34.02 \, \text{g/mol} \)
Step 3: Find mass using \( \text{Mass} = \text{Moles} \times \text{Molar mass} \)
\( \text{Mass} = 2.01 \, \text{mol} \times 34.02 \, \text{g/mol} \approx 68.4 \, \text{g} \)
Step 1: Calculate molar mass of \( \ce{CuBr_2} \)
Molar mass of \( \ce{Cu} = 63.55 \, \text{g/mol} \), \( \ce{Br} = 79.90 \, \text{g/mol} \).
\( \text{Molar mass} = 63.55 + 2(79.90) = 63.55 + 159.80 = 223.35 \, \text{g/mol} \)
Step 2: Find moles of \( \ce{CuBr_2} \)
\( \text{Moles} = \frac{\text{Mass}}{\text{Molar mass}} = \frac{10.0 \, \text{g}}{223.35 \, \text{g/mol}} \approx 0.04477 \, \text{mol} \)
Step 3: Find formula units using Avogadro's number
\( \text{Formula units} = \text{Moles} \times 6.022 \times 10^{23} \)
\( \text{Formula units} = 0.04477 \times 6.022 \times 10^{23} \approx 2.696 \times 10^{22} \approx 2.70 \times 10^{22} \)
Step 1: Balance Mn
Left: 1 Mn, Right: 1 Mn (balanced).
Step 2: Balance I
Right: 3 I (in \( \ce{MnI_3} \)), so \( \ce{HI} \) needs coefficient 3.
Equation: \( \ce{Mn + 3HI -> H_2 + MnI_3} \)
Step 3: Balance H
Left: 3 H, Right: 2 H (in \( \ce{H_2} \)). Multiply \( \ce{H_2} \) by \( \frac{3}{2} \), but use integers. Multiply all coefficients by 2:
\( \ce{2Mn + 6HI -> 3H_2 + 2MnI_3} \)
Verify:
- Mn: 2 left, 2 right.
- H: 6 left, \( 3 \times 2 = 6 \) right.
- I: \( 6 \times 1 = 6 \) left, \( 2 \times 3 = 6 \) right.
Balanced equation: \( \boldsymbol{2\ce{Mn} + 6\ce{HI} -> 3\ce{H_2} + 2\ce{MnI_3}} \)
(Repeat similar steps for other equations, e.g., \( \ce{CaF_2 + Li_2SO_4 -> CaSO_4 + LiF} \)):
- Balance Ca: 1 left, 1 right.
- Balance SO₄: 1 left, 1 right.
- Balance F: 2 left, so \( \ce{LiF} \) needs 2. Then \( \ce{Li_2SO_4} \) needs 1 (to balance Li: \( 2 \times 1 = 2 \) Li, \( \ce{LiF} \) has 2 Li).
Balanced: \( \boldsymbol{\ce{CaF_2} + \ce{Li_2SO_4} -> \ce{CaSO_4} + 2\ce{LiF}} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \approx 68.4 \, \text{g} \) (or more precisely, \( \approx 68.3 \, \text{g} \) if using more precise Avogadro's number)