QUESTION IMAGE
Question
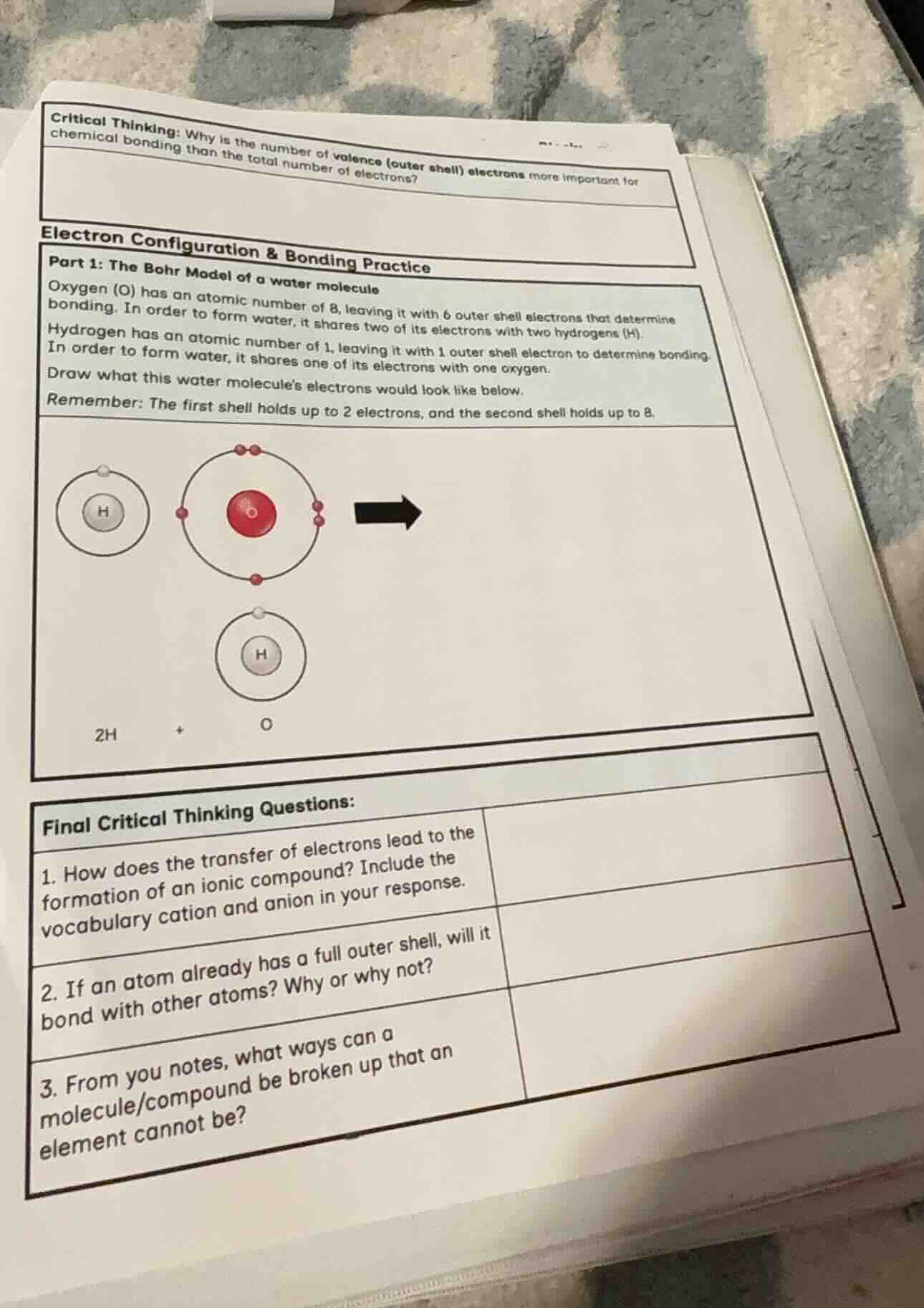
critical thinking: why is the number of valence outer shell electrons more important for chemical bonding than the total number of electrons?electron configuration & bonding practicepart 1: the bohr model of a water moleculeoxygen o has an atomic number of 8, leaving it with 6 outer shell electrons that determine bonding. in order to form water, it shares two of its electrons with two hydrogens h.hydrogen has an atomic number of 1, leaving it with 1 outer shell electron to determine bonding. in order to form water, it shares one of its electrons with one oxygen.draw what this water molecules electrons would look like below.remember: the first shell holds up to 2 electrons, and the second shell holds up to 8.final critical thinking questions:1. how does the transfer of electrons lead to the formation of an ionic compound? include the vocabulary cation and anion in your response.2. if an atom already has a full outer shell, will it bond with other atoms? why or why not?3. from you notes, what ways can a molecule/compound be broken up that an element cannot be?
First: Bohr Model of Water Molecule
Step1: Fulfill H's outer shell
Hydrogen (1 proton, 1 valence electron) shares its electron with oxygen, and gains a shared electron from oxygen to fill its 1st shell (max 2 electrons).
Step2: Fulfill O's outer shell
Oxygen (8 protons, 6 valence electrons) shares 1 electron with each hydrogen, giving it 8 total outer-shell electrons (6 own + 2 shared) to fill its 2nd shell (max 8 electrons).
Step3: Draw the bonded molecule
The final structure has a central oxygen atom with a full outer shell (8 electrons: 4 unshared, 2 pairs shared with H), and each hydrogen atom with a full outer shell (2 electrons: 1 own, 1 shared with O).
- Ionic compounds form when electrons transfer between atoms: a metal loses valence electrons to become a positively charged cation, and a nonmetal gains those electrons to become a negatively charged anion. The opposite electric charges of the cation and anion attract strongly, forming a stable ionic bond and ionic compound.
- An atom with a full outer shell will not bond with other atoms. Full outer electron shells mean the atom is already in a low-energy, stable state, so it has no drive to gain, lose, or share electrons to achieve stability.
- Molecules/compounds can be broken down via chemical reactions (e.g., hydrolysis of water into H₂ and O₂, or decomposition of salt via electrolysis). Elements cannot be broken down further by chemical means, as they consist of only one type of atom.
- Valence electrons are more important for bonding because only outer-shell electrons are involved in gaining, losing, or sharing to achieve a full, stable outer electron configuration. Total electrons include inner-shell electrons, which are already stable and do not participate in bonding interactions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Visual description of the Bohr model: A central red oxygen nucleus, surrounded by a second electron shell with 8 electrons (4 as unpaired dots, 2 pairs each connected to a hydrogen nucleus). Each hydrogen nucleus has a single electron shell with 2 electrons (one original, one shared from oxygen) orbiting it, connected to the oxygen shell.)
---