QUESTION IMAGE
Question
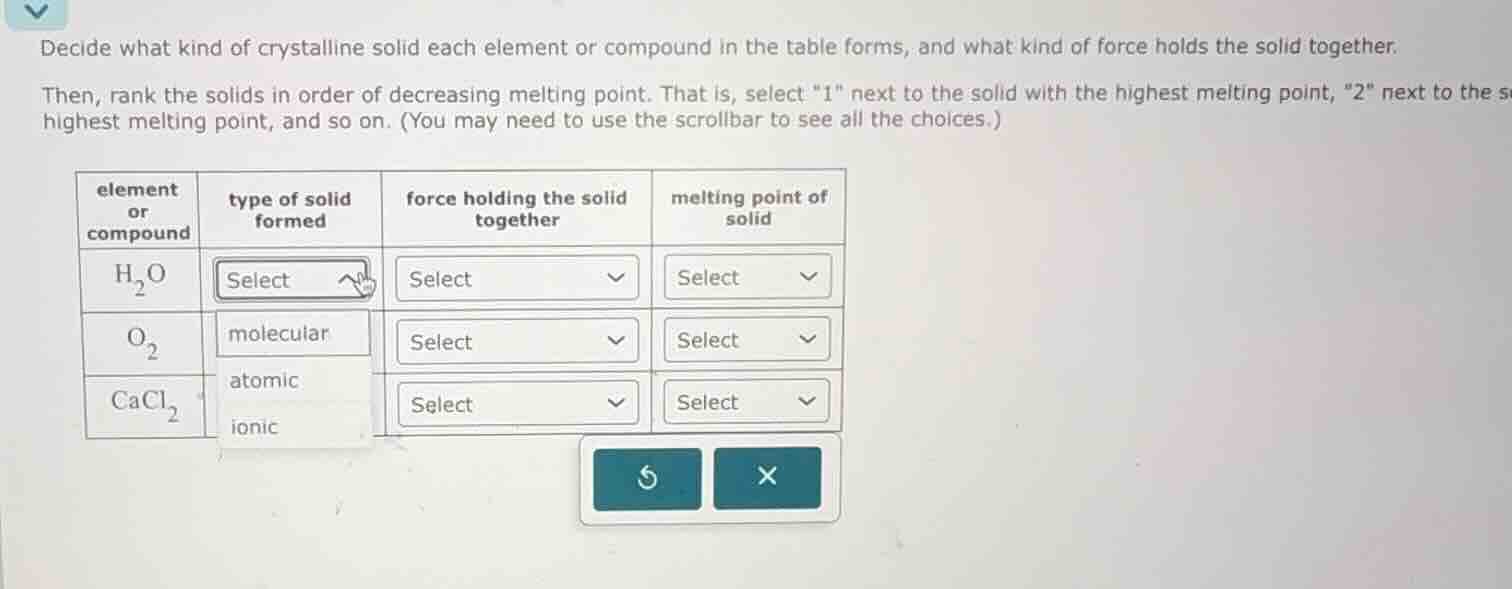
decide what kind of crystalline solid each element or compound in the table forms, and what kind of force holds the solid together. then, rank the solids in order of decreasing melting point. that is, select \1\ next to the solid with the highest melting point, \2\ next to the s highest melting point, and so on. (you may need to use the scrollbar to see all the choices.)
For $\boldsymbol{\ce{H2O}}$:
Step 1: Determine the type of solid
$\ce{H2O}$ is a covalent compound composed of molecules (each $\ce{H2O}$ is a molecule), so it forms a molecular solid.
Step 2: Determine the force holding the solid together
In molecular solids, the forces holding the molecules together are intermolecular forces. For $\ce{H2O}$, the dominant intermolecular force is hydrogen bonding (a special type of dipole - dipole interaction, but stronger, due to the presence of highly electronegative O bonded to H).
Step 3: Recall the melting point of $\ce{H2O}$
The melting point of $\ce{H2O}$ (ice) is $0^\circ\mathrm{C}$ (or $273.15\mathrm{K}$).
For $\boldsymbol{\ce{O2}}$:
Step 1: Determine the type of solid (already given as molecular)
$\ce{O2}$ consists of $\ce{O2}$ molecules, so it is a molecular solid.
Step 2: Determine the force holding the solid together
For non - polar molecular solids like $\ce{O2}$, the intermolecular force is London dispersion forces (also called van der Waals forces). These are weak forces resulting from temporary dipoles in the molecules.
Step 3: Recall the melting point of $\ce{O2}$
The melting point of $\ce{O2}$ is approximately $-218^\circ\mathrm{C}$ (or $55.15\mathrm{K}$).
For $\boldsymbol{\ce{CaCl2}}$:
Step 1: Determine the type of solid
$\ce{CaCl2}$ is composed of $\ce{Ca^{2+}}$ and $\ce{Cl^-}$ ions. So, it forms an ionic solid.
Step 2: Determine the force holding the solid together
In ionic solids, the force holding the ions together is the ionic bond (electrostatic attraction between positively charged cations and negatively charged anions).
Step 3: Recall the melting point of $\ce{CaCl2}$
The melting point of $\ce{CaCl2}$ is approximately $772^\circ\mathrm{C}$ (or $1045.15\mathrm{K}$).
Ranking in order of decreasing melting point:
Ionic solids generally have higher melting points than molecular solids because ionic bonds are much stronger than intermolecular forces. Among molecular solids, substances with stronger intermolecular forces have higher melting points. $\ce{CaCl2}$ (ionic, melting point ~$772^\circ\mathrm{C}$) has a higher melting point than $\ce{H2O}$ (molecular, melting point $0^\circ\mathrm{C}$), and $\ce{H2O}$ has a higher melting point than $\ce{O2}$ (molecular, melting point ~$-218^\circ\mathrm{C}$). So the order from highest to lowest melting point is: $\ce{CaCl2}$ (rank 1), $\ce{H2O}$ (rank 2), $\ce{O2}$ (rank 3).
Filling the table:
| element or compound | type of solid formed | force holding the solid together | melting point of solid | rank (decreasing mp) |
|---|---|---|---|---|
| $\ce{O2}$ | molecular | London dispersion forces | $-218^\circ\mathrm{C}$ | 3 |
| $\ce{CaCl2}$ | ionic | ionic bond | $772^\circ\mathrm{C}$ | 1 |
Final Answers (for the table fields):
- $\ce{H2O}$: type of solid - molecular; force - hydrogen bonding; melting point - $0^\circ\mathrm{C}$; rank - 2
- $\ce{O2}$: type of solid - molecular; force - London dispersion forces; melting point - $-218^\circ\mathrm{C}$; rank - 3
- $\ce{CaCl2}$: type of solid - ionic; force - ionic bond; melting point - $772^\circ\mathrm{C}$; rank - 1
(If you only need the type of solid, force, and melting point for each compound as per the table dropdowns, the above details can be used to fill them. For ranking, the order of decreasing melting point is $\ce{CaCl2}$ (1), $\ce{H2O}$ (2), $\ce{O2}$ (3))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For $\boldsymbol{\ce{H2O}}$:
Step 1: Determine the type of solid
$\ce{H2O}$ is a covalent compound composed of molecules (each $\ce{H2O}$ is a molecule), so it forms a molecular solid.
Step 2: Determine the force holding the solid together
In molecular solids, the forces holding the molecules together are intermolecular forces. For $\ce{H2O}$, the dominant intermolecular force is hydrogen bonding (a special type of dipole - dipole interaction, but stronger, due to the presence of highly electronegative O bonded to H).
Step 3: Recall the melting point of $\ce{H2O}$
The melting point of $\ce{H2O}$ (ice) is $0^\circ\mathrm{C}$ (or $273.15\mathrm{K}$).
For $\boldsymbol{\ce{O2}}$:
Step 1: Determine the type of solid (already given as molecular)
$\ce{O2}$ consists of $\ce{O2}$ molecules, so it is a molecular solid.
Step 2: Determine the force holding the solid together
For non - polar molecular solids like $\ce{O2}$, the intermolecular force is London dispersion forces (also called van der Waals forces). These are weak forces resulting from temporary dipoles in the molecules.
Step 3: Recall the melting point of $\ce{O2}$
The melting point of $\ce{O2}$ is approximately $-218^\circ\mathrm{C}$ (or $55.15\mathrm{K}$).
For $\boldsymbol{\ce{CaCl2}}$:
Step 1: Determine the type of solid
$\ce{CaCl2}$ is composed of $\ce{Ca^{2+}}$ and $\ce{Cl^-}$ ions. So, it forms an ionic solid.
Step 2: Determine the force holding the solid together
In ionic solids, the force holding the ions together is the ionic bond (electrostatic attraction between positively charged cations and negatively charged anions).
Step 3: Recall the melting point of $\ce{CaCl2}$
The melting point of $\ce{CaCl2}$ is approximately $772^\circ\mathrm{C}$ (or $1045.15\mathrm{K}$).
Ranking in order of decreasing melting point:
Ionic solids generally have higher melting points than molecular solids because ionic bonds are much stronger than intermolecular forces. Among molecular solids, substances with stronger intermolecular forces have higher melting points. $\ce{CaCl2}$ (ionic, melting point ~$772^\circ\mathrm{C}$) has a higher melting point than $\ce{H2O}$ (molecular, melting point $0^\circ\mathrm{C}$), and $\ce{H2O}$ has a higher melting point than $\ce{O2}$ (molecular, melting point ~$-218^\circ\mathrm{C}$). So the order from highest to lowest melting point is: $\ce{CaCl2}$ (rank 1), $\ce{H2O}$ (rank 2), $\ce{O2}$ (rank 3).
Filling the table:
| element or compound | type of solid formed | force holding the solid together | melting point of solid | rank (decreasing mp) |
|---|---|---|---|---|
| $\ce{O2}$ | molecular | London dispersion forces | $-218^\circ\mathrm{C}$ | 3 |
| $\ce{CaCl2}$ | ionic | ionic bond | $772^\circ\mathrm{C}$ | 1 |
Final Answers (for the table fields):
- $\ce{H2O}$: type of solid - molecular; force - hydrogen bonding; melting point - $0^\circ\mathrm{C}$; rank - 2
- $\ce{O2}$: type of solid - molecular; force - London dispersion forces; melting point - $-218^\circ\mathrm{C}$; rank - 3
- $\ce{CaCl2}$: type of solid - ionic; force - ionic bond; melting point - $772^\circ\mathrm{C}$; rank - 1
(If you only need the type of solid, force, and melting point for each compound as per the table dropdowns, the above details can be used to fill them. For ranking, the order of decreasing melting point is $\ce{CaCl2}$ (1), $\ce{H2O}$ (2), $\ce{O2}$ (3))