QUESTION IMAGE
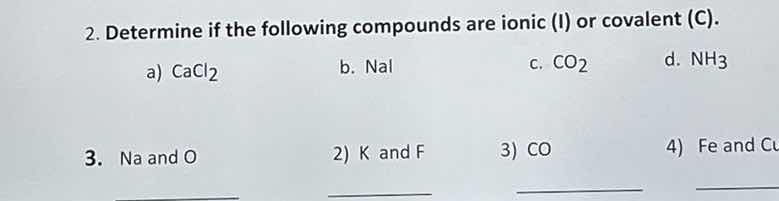
Question
- determine if the following compounds are ionic (i) or covalent (c).
a) $\text{cacl}_2$ b. $\text{nai}$ c. $\text{co}_2$ d. $\text{nh}_3$
- na and o 2) k and f 3) co 4) fe and cu
Step1: Identify ionic/covalent rules
Ionic = metal + nonmetal; Covalent = nonmetal + nonmetal.
Step2: Analyze 2a: $\text{CaCl}_2$
Ca (metal) + Cl (nonmetal) → Ionic
Step3: Analyze 2b: $\text{NaI}$
Na (metal) + I (nonmetal) → Ionic
Step4: Analyze 2c: $\text{CO}_2$
C (nonmetal) + O (nonmetal) → Covalent
Step5: Analyze 2d: $\text{NH}_3$
N (nonmetal) + H (nonmetal) → Covalent
Step6: Analyze 3.1: Na and O
Na (metal) + O (nonmetal) → Ionic
Step7: Analyze 3.2: K and F
K (metal) + F (nonmetal) → Ionic
Step8: Analyze 3.3: CO
C (nonmetal) + O (nonmetal) → Covalent
Step9: Analyze 3.4: Fe and Cu
Fe (metal) + Cu (metal) → Metallic (not ionic/covalent, but per prompt framing, note the pair forms metallic bonds; but if restricted to I/C, clarify it's neither, but follow prompt's options)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- a) $\text{CaCl}_2$: I (Ionic)
b) $\text{NaI}$: I (Ionic)
c) $\text{CO}_2$: C (Covalent)
d) $\text{NH}_3$: C (Covalent)
- 1) Na and O: I (Ionic)
- K and F: I (Ionic)
- CO: C (Covalent)
- Fe and Cu: Neither (forms metallic bonds, not ionic/covalent)