QUESTION IMAGE
Question
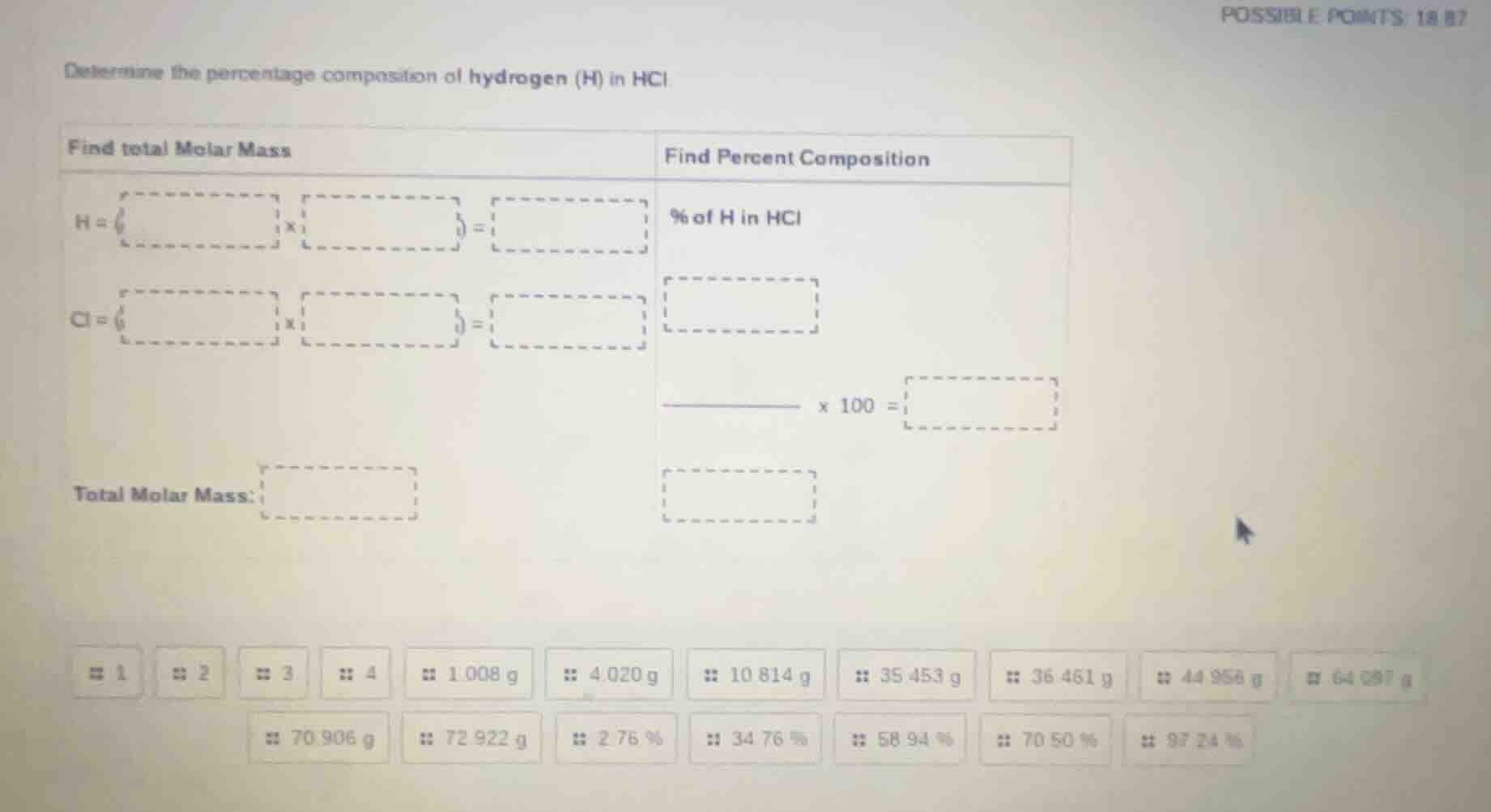
determine the percentage composition of hydrogen (h) in hcl
find total molar mass\tfind percent composition
h = ( ) x ( ) = \t% of h in hcl
cl = ( ) x ( ) = \t
\t
total molar mass: \t
1 2 3 4 1.008 g 4.020 g 10.814 g 35.453 g 36.461 g 44.956 g 64.087 g
70.906 g 72.922 g 2.76 % 34.76 % 58.94 % 70.50 % 97.24 %
Step1: Calculate molar mass of H
Hydrogen (H) has 1 atom, and atomic mass of H is 1.008 g. So molar mass of H: $1\times1.008\ \text{g} = 1.008\ \text{g}$
Step2: Calculate molar mass of Cl
Chlorine (Cl) has 1 atom, atomic mass of Cl is 35.453 g. So molar mass of Cl: $1\times35.453\ \text{g} = 35.453\ \text{g}$
Step3: Calculate total molar mass of HCl
Total molar mass = molar mass of H + molar mass of Cl = $1.008\ \text{g} + 35.453\ \text{g} = 36.461\ \text{g}$
Step4: Calculate percentage composition of H
Percentage composition of H = $\frac{\text{Molar mass of H}}{\text{Total molar mass of HCl}}\times100$ = $\frac{1.008\ \text{g}}{36.461\ \text{g}}\times100 \approx 2.76\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The percentage composition of hydrogen (H) in HCl is approximately 2.76%.