QUESTION IMAGE
Question
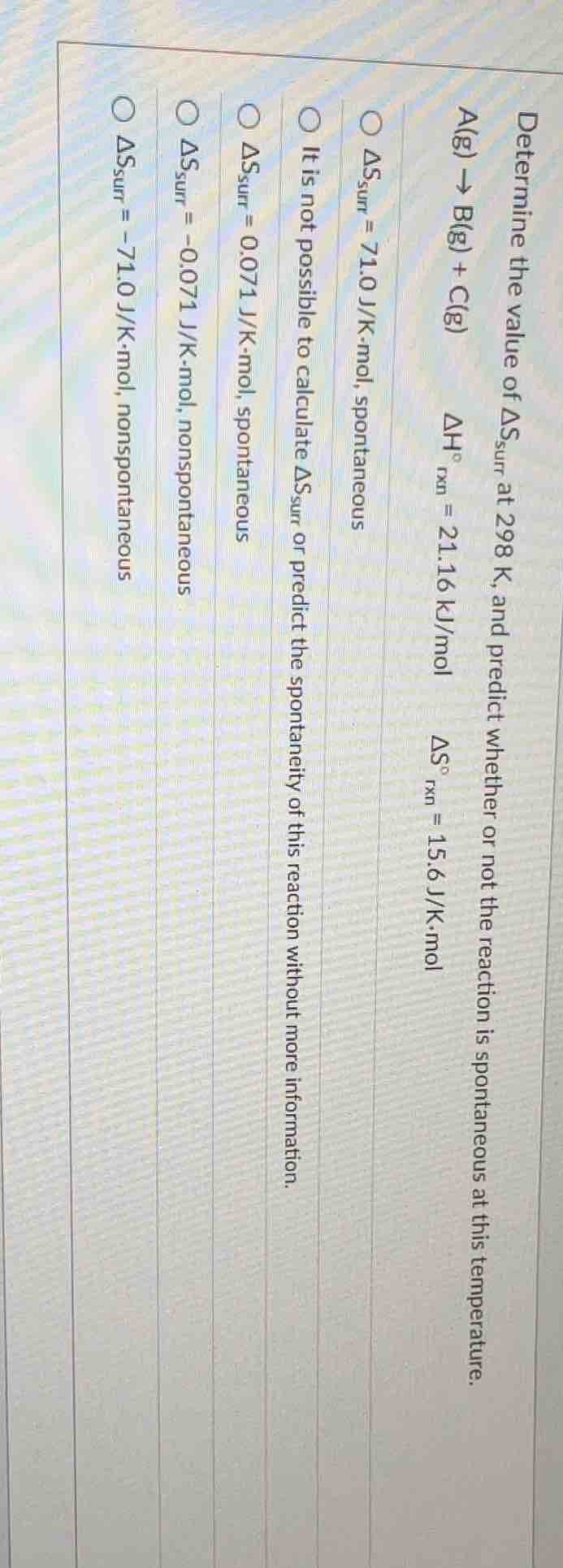
determine the value of $\delta s_{sur}$ at 298 k, and predict whether or not the reaction is spontaneous at this temperature.\
$\ce{a(g) \
ightarrow b(g) + c(g)}$\
$\delta h^\circ_{rxn} = 21.16$ kj/mol\
$\delta s^\circ_{rxn} = 15.6$ j/k·mol\
\
\\(\delta s_{sur} = 71.0\\) j/k·mol, spontaneous\
it is not possible to calculate \\(\delta s_{sur}\\) or predict the spontaneity of this reaction without more information.\
\\(\delta s_{sur} = 0.071\\) j/k·mol, spontaneous\
\\(\delta s_{sur} = -0.071\\) j/k·mol, nonspontaneous\
\\(\delta s_{sur} = -71.0\\) j/k·mol, nonspontaneous
Step1: Recall the formula for entropy of surroundings
The formula for the entropy change of the surroundings ($\Delta S_{\text{sur}}$) is $\Delta S_{\text{sur}} = \frac{-\Delta H_{\text{rxn}}^{\circ}}{T}$, where $\Delta H_{\text{rxn}}^{\circ}$ is the enthalpy change of the reaction and $T$ is the temperature in Kelvin.
Step2: Convert $\Delta H_{\text{rxn}}^{\circ}$ to J/mol
Given $\Delta H_{\text{rxn}}^{\circ} = 21.16\ \text{kJ/mol}$, convert it to J/mol: $21.16\ \text{kJ/mol} = 21160\ \text{J/mol}$.
Step3: Calculate $\Delta S_{\text{sur}}$
Substitute $\Delta H_{\text{rxn}}^{\circ} = 21160\ \text{J/mol}$ and $T = 298\ \text{K}$ into the formula:
$\Delta S_{\text{sur}} = \frac{-21160\ \text{J/mol}}{298\ \text{K}} \approx -71.0\ \text{J/K·mol}$.
Step4: Determine spontaneity
To determine spontaneity, we can check the Gibbs free energy change $\Delta G = \Delta H - T\Delta S$. First, calculate $T\Delta S$: $T\Delta S_{\text{rxn}}^{\circ} = 298\ \text{K} \times 15.6\ \text{J/K·mol} = 4648.8\ \text{J/mol} = 4.6488\ \text{kJ/mol}$. Then $\Delta G = \Delta H - T\Delta S = 21.16\ \text{kJ/mol} - 4.6488\ \text{kJ/mol} \approx 16.51\ \text{kJ/mol} > 0$, so the reaction is nonspontaneous.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\Delta S_{\text{sur}} = -71.0\ \text{J/K·mol}$, nonspontaneous